Scientists at Cedars-Sinai Medical Center report that a subset of astrocytes located away from a spinal cord injury can help drive repair in mice by releasing the protein CCN1, which alters microglia metabolism to improve cleanup of lipid-rich nerve debris. The work, published in Nature, also found evidence of a similar CCN1-linked response in human spinal cord tissue from people with multiple sclerosis.
Neuroscientist Joshua Burda and colleagues at Cedars-Sinai investigated how the spinal cord responds to traumatic injury in mice, focusing on how damage triggers inflammation and affects tissue repair.
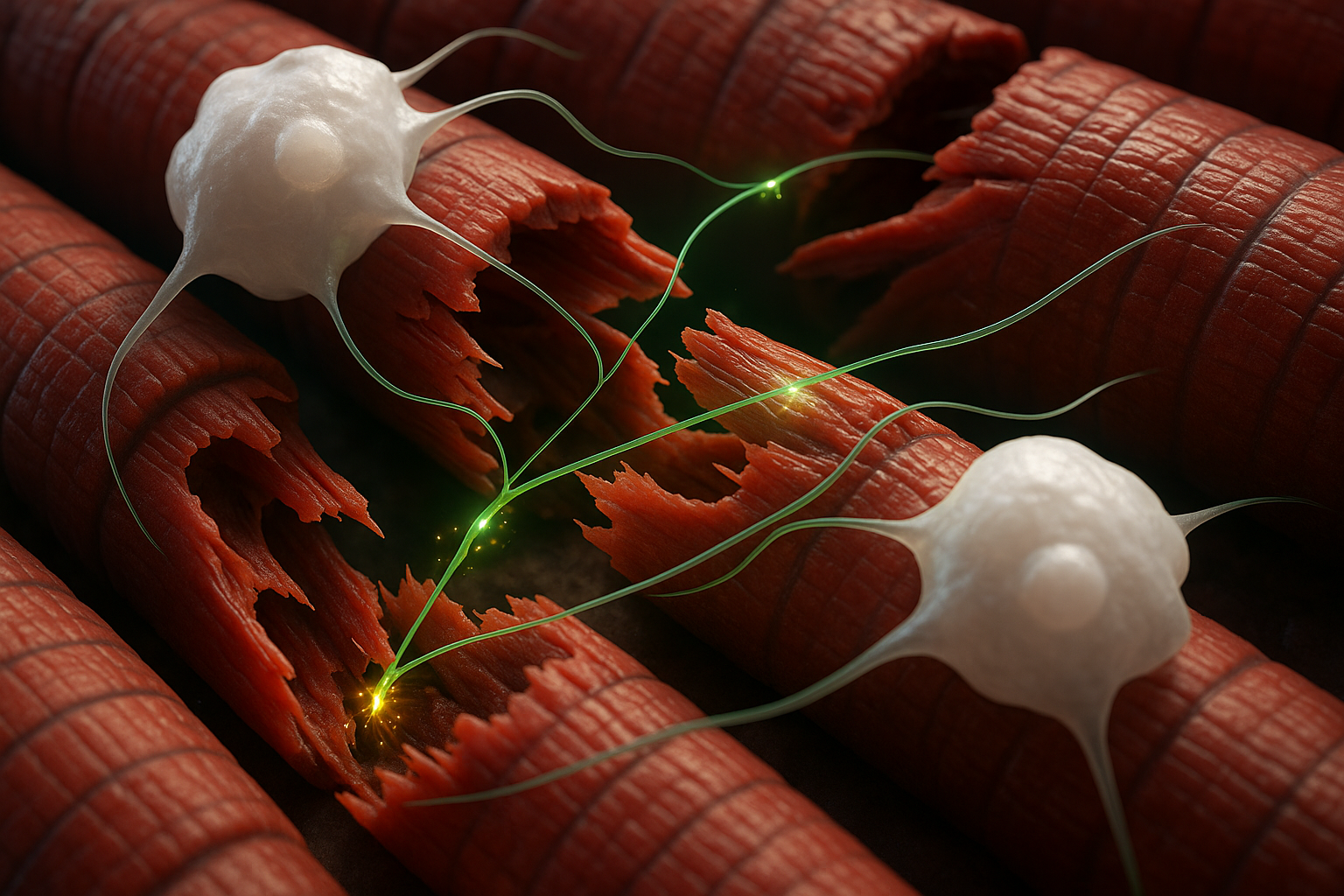
The spinal cord is a long bundle of nerve tissue that runs from the brain down the back. It contains an inner gray-matter region with nerve cell bodies and support cells called astrocytes, and an outer white-matter region made up largely of long nerve fibers and astrocytes. When the spinal cord is injured, nerve fibers can be torn, which may cause paralysis and disrupt sensations such as touch and temperature. The damaged fibers break down into debris, and because nerve fibers extend over long distances, inflammation and degeneration can spread beyond the original injury site.
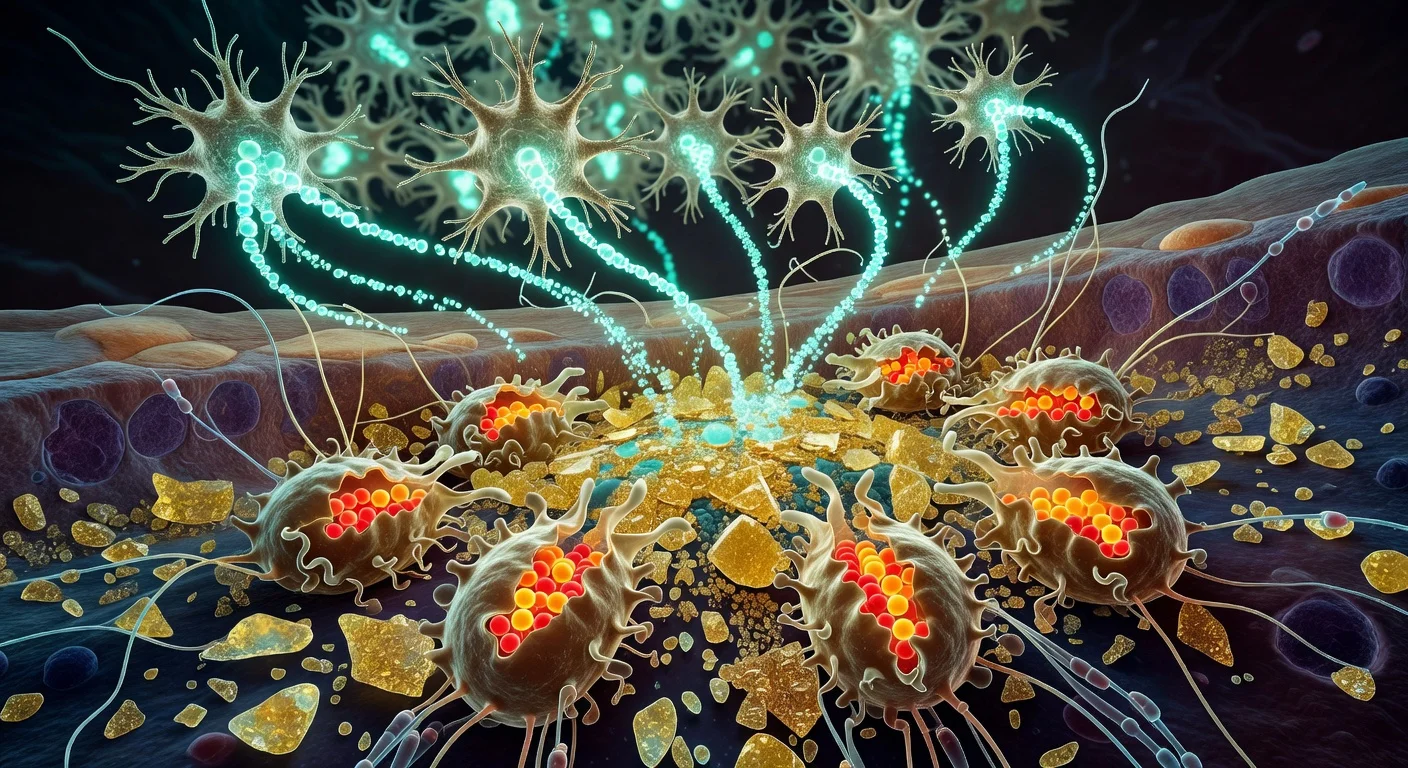
In mouse experiments, the team identified astrocytes located away from the injury that become reactive after damage. The researchers called these cells “lesion-remote astrocytes” (LRAs) and reported that LRAs include multiple subtypes.
One LRA subtype, the study found, produces the secreted protein CCN1 (also known as CYR61). Burda said CCN1 helps signal microglia—immune cells in the central nervous system—to handle the lipid-rich debris produced when nerve fibers and myelin break down.
“One function of microglia is to serve as chief garbage collectors in the central nervous system,” Burda said. “After tissue damage, they eat up pieces of nerve fiber debris—which are very fatty and can cause them to get a kind of indigestion. Our experiments showed that astrocyte CCN1 signals the microglia to change their metabolism so they can better digest all that fat.”
The Nature paper reports that when astrocyte-derived CCN1 was removed in the mouse models, debris clearance was impaired and microglia showed abnormal activation and disrupted lipid handling, with increased clustering of debris-laden microglia and reduced measures of repair and neurological recovery.
The researchers also examined human spinal cord tissue from people with multiple sclerosis and reported evidence consistent with a CCN1-associated astrocyte response in areas of myelin damage, suggesting that related repair programs may also be engaged in demyelinating disease.
“The role of astrocytes in central nervous system healing is remarkably understudied,” said David Underhill, chair of Cedars-Sinai’s Department of Biomedical Sciences. “This work strongly suggests that lesion-remote astrocytes offer a viable path for limiting chronic inflammation, enhancing functionally meaningful regeneration, and promoting neurological recovery after brain and spinal cord injury and in disease.”
Burda’s group said it is now working on strategies aimed at harnessing the CCN1 pathway to improve spinal cord healing, while also exploring whether similar mechanisms may be relevant to other central nervous system injuries and disorders.
The research was supported by grants from the U.S. National Institutes of Health and other funders, including the Paralyzed Veterans Research Foundation of America and Wings for Life.