Researchers in France have found that hormone‑sensitive lipase (HSL), long known for breaking down stored fat, also operates in the nucleus of fat cells to help maintain adipose tissue health. When HSL is missing, fat tissue in mice shrinks instead of expanding, leading to lipodystrophy, a finding that helps explain shared health risks between obesity and fat‑loss disorders.
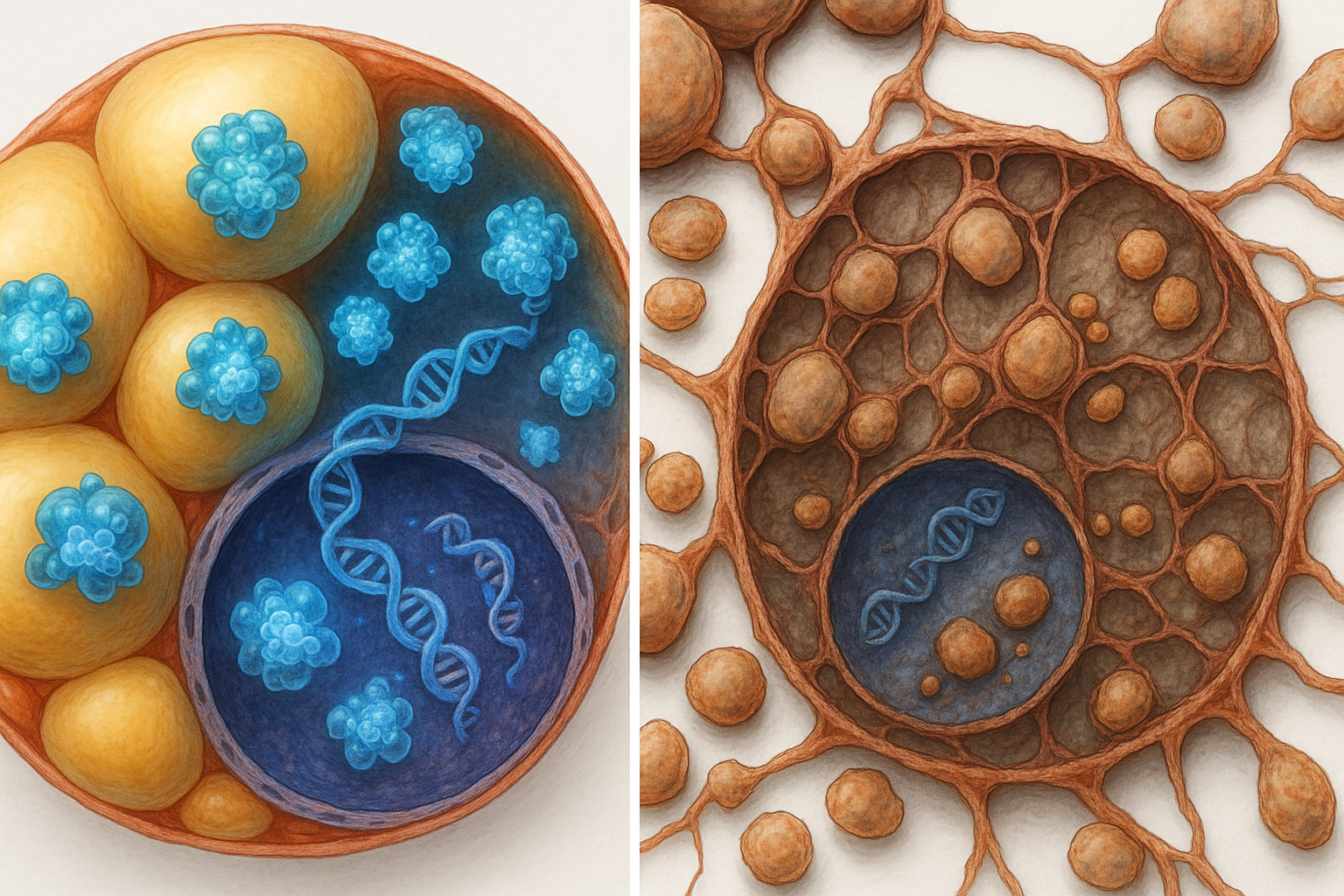
Fat cells, or adipocytes, store energy in lipid droplets, which the body can draw on during times of need, such as fasting. Hormone‑sensitive lipase (HSL) has been known for decades as a key lipase in adipocytes: when activated by hormones such as catecholamines, it helps break down stored fat, releasing energy for use by organs.
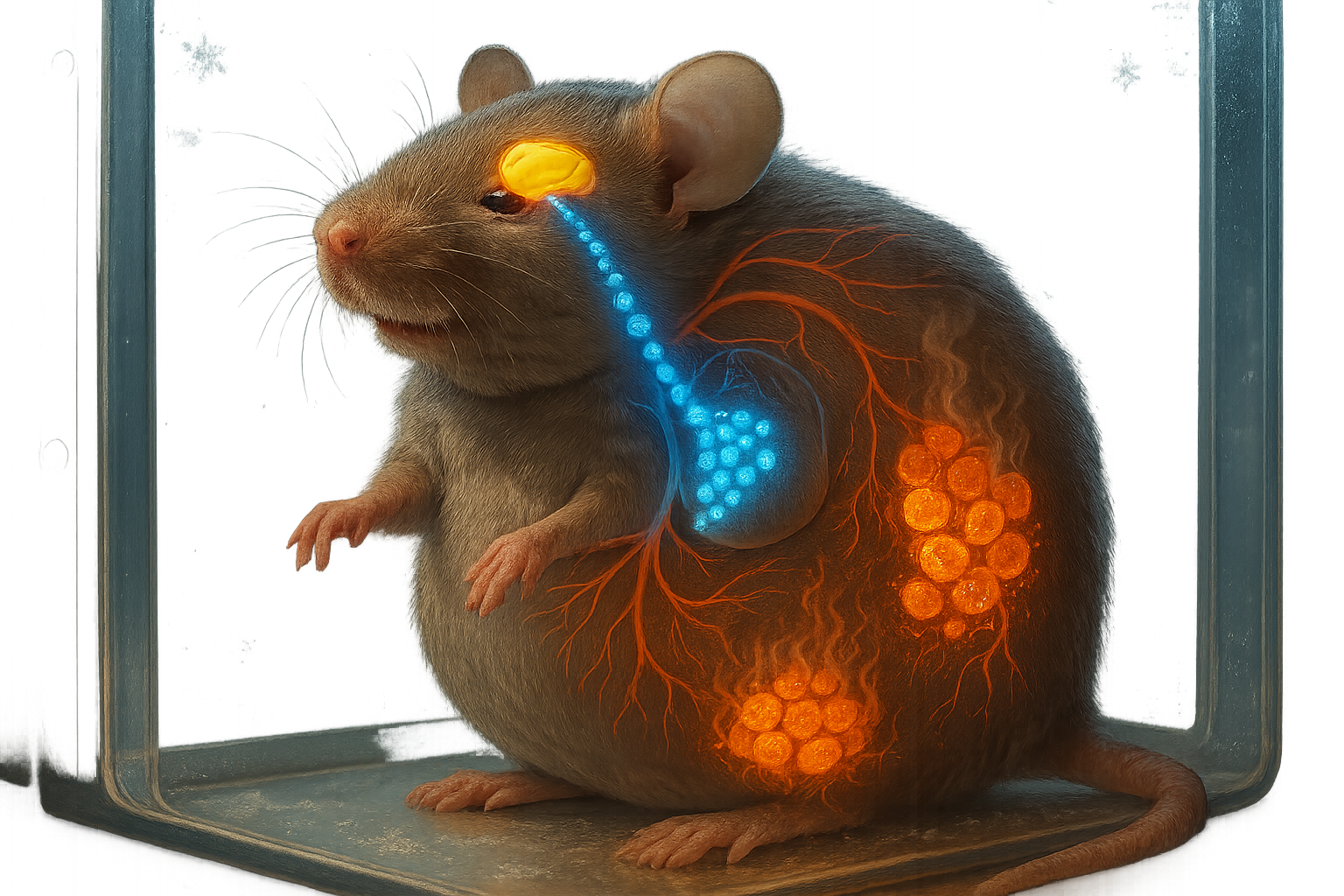
Yet studies in mice and in patients with mutations in the HSL gene have revealed a paradoxical outcome. Instead of developing excessive fat accumulation and obesity, animals and people lacking functional HSL show a marked reduction in fat mass and features of lipodystrophy, a condition in which adipose tissue is abnormally reduced or dysfunctional. (sciencedaily.com)
To probe this paradox, a team led by Dominique Langin, a professor at the University of Toulouse's Institute of Metabolic and Cardiovascular Diseases (I2MC), examined where HSL is located inside adipocytes. In addition to its established presence at the surface of lipid droplets, where it participates in fat breakdown, the researchers found that HSL is also present in the nucleus of fat cells. "In the nucleus of adipocytes, HSL is able to associate with many other proteins and take part in a program that maintains an optimal amount of adipose tissue and keeps adipocytes 'healthy'," said co‑author Jérémy Dufau, who completed his doctoral thesis on this topic. (sciencedaily.com)
The amount of HSL in the nucleus appears to be dynamically regulated. According to the release from Université de Toulouse, adrenaline, which activates HSL on lipid droplets, also promotes its exit from the nucleus, a process that occurs during fasting. In obese mice, by contrast, nuclear HSL levels are increased, suggesting that the normal balance of HSL between the nucleus and lipid droplets is disturbed in obesity. (sciencedaily.com)
"HSL has been known since the 1960s as a fat‑mobilizing enzyme. But we now know that it also plays an essential role in the nucleus of adipocytes, where it helps maintain healthy adipose tissue," Langin said, as quoted by Université de Toulouse and related summaries of the work. This dual role offers a possible explanation for why both obesity, characterized by excessive fat, and lipodystrophy, marked by a lack of functional fat, can lead to malfunctioning adipocytes and similar metabolic and cardiovascular complications. (eurekalert.org)
The work, reported by Université de Toulouse and other outlets summarizing a study in Cell Metabolism, comes at a time of rising obesity rates worldwide. The ScienceDaily summary, drawing on data from global health agencies, notes that billions of people are now affected by overweight or obesity. The discovery of a nuclear regulatory role for HSL adds a new layer to scientists' understanding of metabolic disease and may ultimately inform future approaches to prevention and treatment, though clinical applications remain preliminary. (sciencedaily.com)