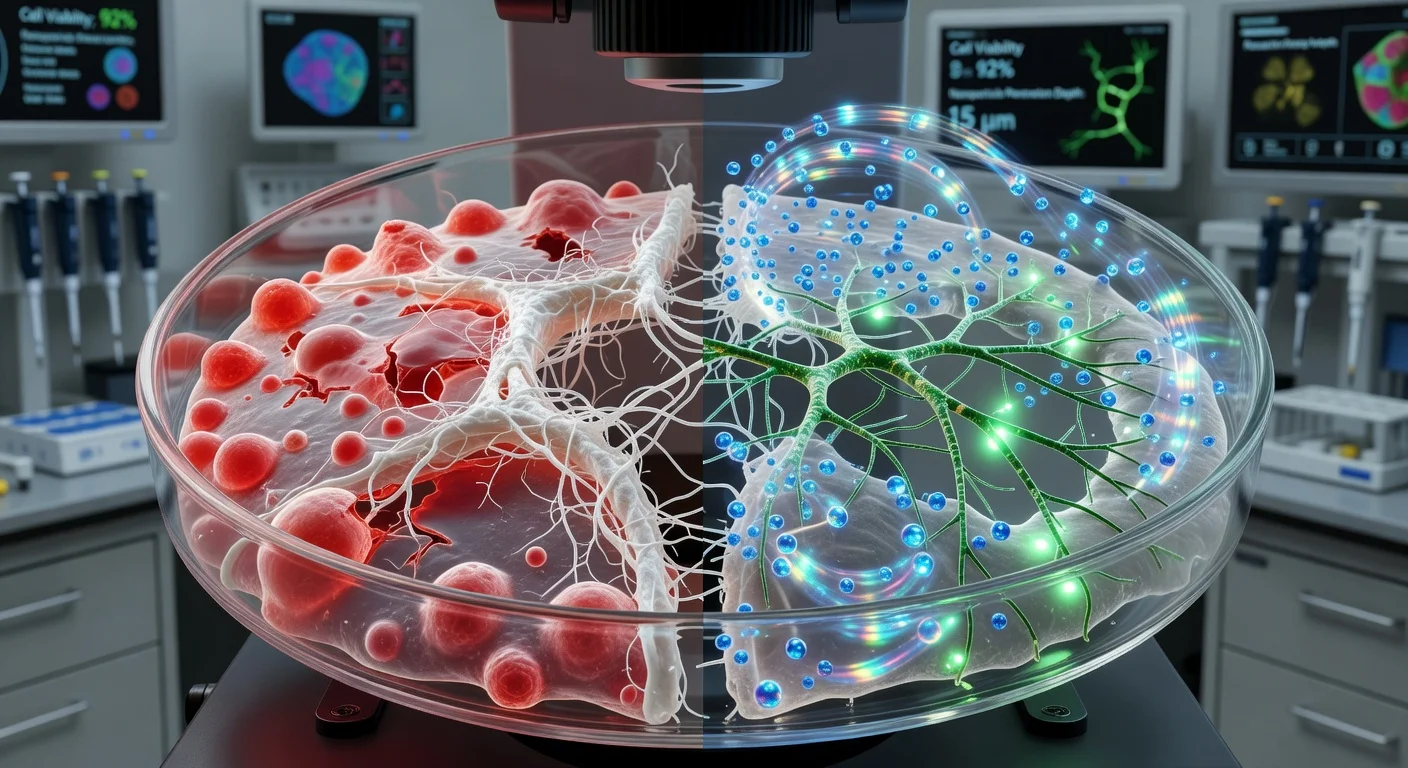
Researchers at Texas A&M University say they have developed a customizable “vessel-chip” that recreates the complex shapes of human blood vessels—including branches, aneurysm-like bulges and stenosis-like narrowings—so scientists can study how altered blood flow affects endothelial cells and evaluate potential treatments without relying on animal models.
Human blood vessels are not uniform tubes: they bend, branch, narrow and widen in ways that change blood flow and are linked to where vascular disease can develop. Researchers in the Department of Biomedical Engineering at Texas A&M University have reported a microfluidic “vessel-chip” platform designed to reproduce that architectural complexity.
The device is a small-scale vessel-on-a-chip system lined with endothelial cells—the cells that form the inner lining of blood vessels—so the team can examine how flow patterns translate into differences in shear stress and endothelial responses. Jennifer D. Lee, identified by Texas A&M as a biomedical engineering master’s student working in the lab of Abhishek Jain, described the motivation as capturing how branching, aneurysm-like expansions and stenosis-like restrictions can substantially alter flow patterns and the stresses experienced by vessel walls.
According to the university’s release, the project builds on earlier work in the same group by Tanmay Mathur, described as a former graduate student who developed a straight vessel-chip design. The paper lists Lee, Ankit Kumar, Mathur and Jain as authors and was published in Lab on a Chip as “Vascular architecture-on-chip: engineering complex blood vessels for reproducing physiological and heterogeneous hemodynamics and endothelial function” (2025, volume 25, issue 11, pages 2620–2631; DOI: 10.1039/D4LC00968A).
Jain, whom Texas A&M describes as an associate professor and the Barbara and Ralph Cox ’53 faculty fellow in biomedical engineering, said the new approach makes it possible to create more complex, living vessel structures and study disease-relevant sites that may be difficult to reproduce with simpler models.
Texas A&M said Lee began the work as an undergraduate honors student seeking hands-on research experience and continued through the university’s Master of Science fast-track program. The release also quoted Lee describing the lab environment as helping her develop collaboration and communication skills.
The team said future iterations may add cell types beyond endothelial cells to study interactions between flowing blood and surrounding tissues—an effort Jain described as progressing toward what he called a “fourth dimensionality” in organs-on-a-chip research.
In the university’s account, the project received support from multiple organizations, including the U.S. Army Medical Research Program, NASA, the Biomedical Advanced Research and Development Authority, the National Institutes of Health, the U.S. Food and Drug Administration, the National Science Foundation, and Texas A&M’s Office of Innovation Translational Investment Funds.
Researchers said the vessel-chip platform could support patient-tailored studies of blood flow and treatment response, offering a non-animal system for evaluating potential therapies and studying vascular disease mechanisms.