Drug recall
Hoton da AI ya samar
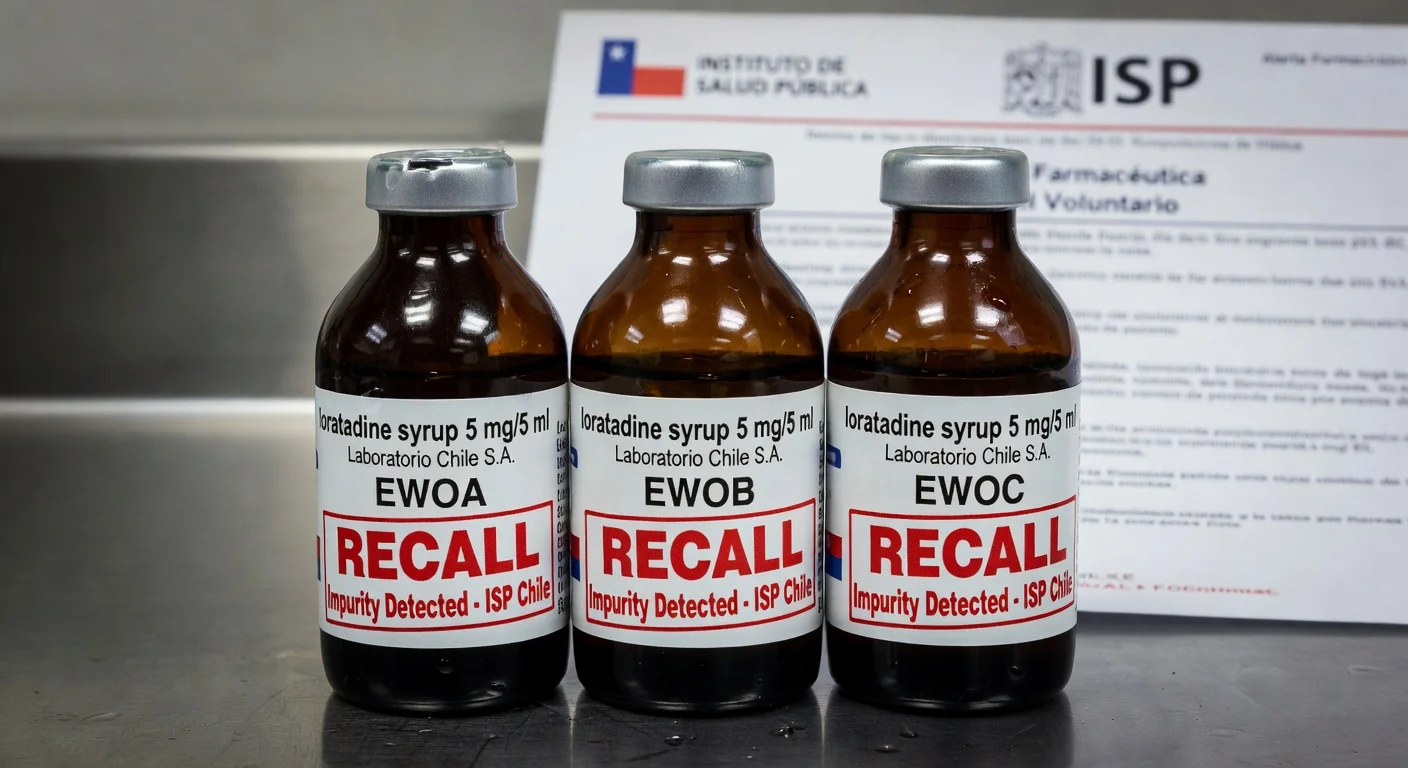
ISP orders voluntary recall of loratadine syrup due to impurities
An Ruwaito ta hanyar AI Hoton da AI ya samar
Chile's Public Health Institute (ISP) issued a pharmaceutical alert ordering the voluntary recall of Laboratorio Chile S.A.'s loratadine syrup 5 mg/5 ml after detecting out-of-specification impurities in stability studies. The action targets batches EWOA, EWOB, and EWOC, expiring in September 2026.