Drug recall
Picha iliyoundwa na AI
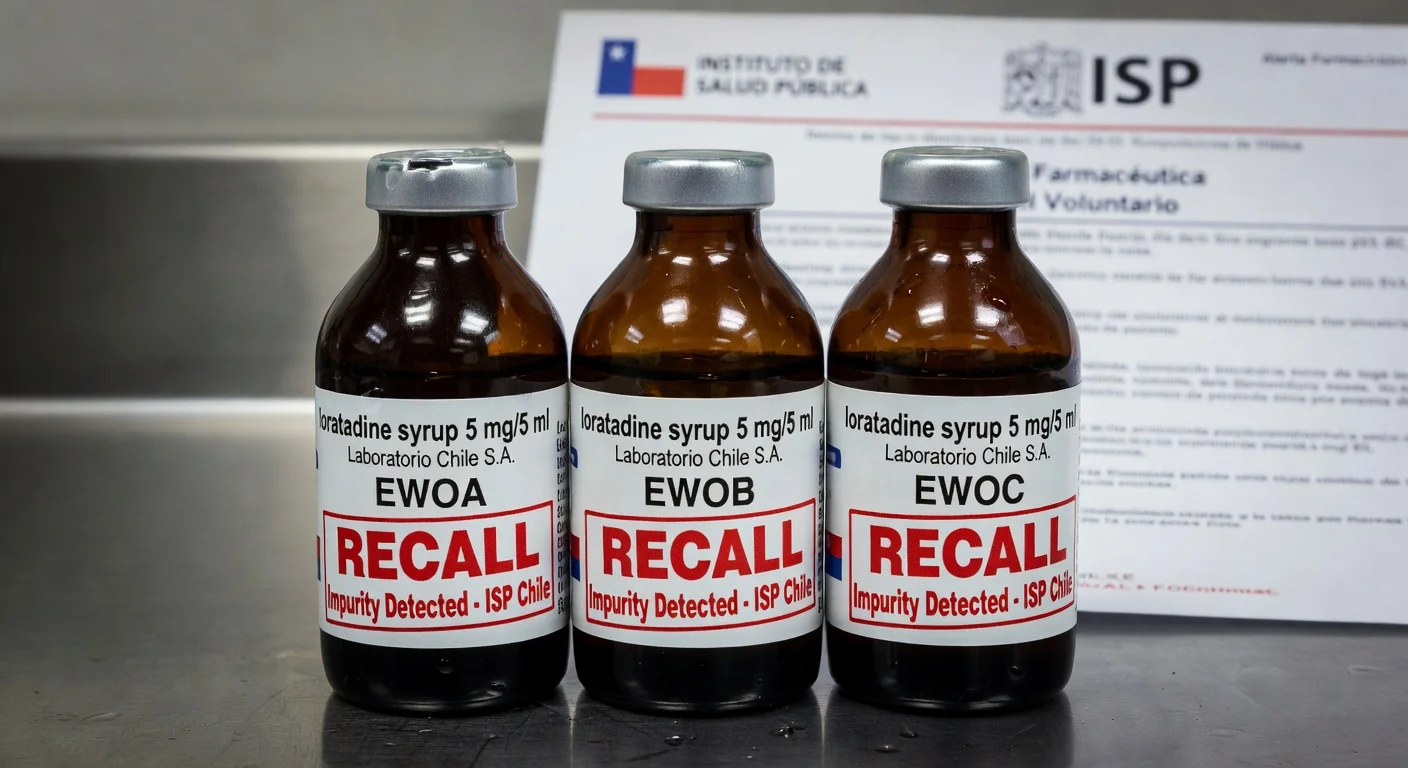
ISP orders voluntary recall of loratadine syrup due to impurities
Imeripotiwa na AI Picha iliyoundwa na AI
Chile's Public Health Institute (ISP) issued a pharmaceutical alert ordering the voluntary recall of Laboratorio Chile S.A.'s loratadine syrup 5 mg/5 ml after detecting out-of-specification impurities in stability studies. The action targets batches EWOA, EWOB, and EWOC, expiring in September 2026.