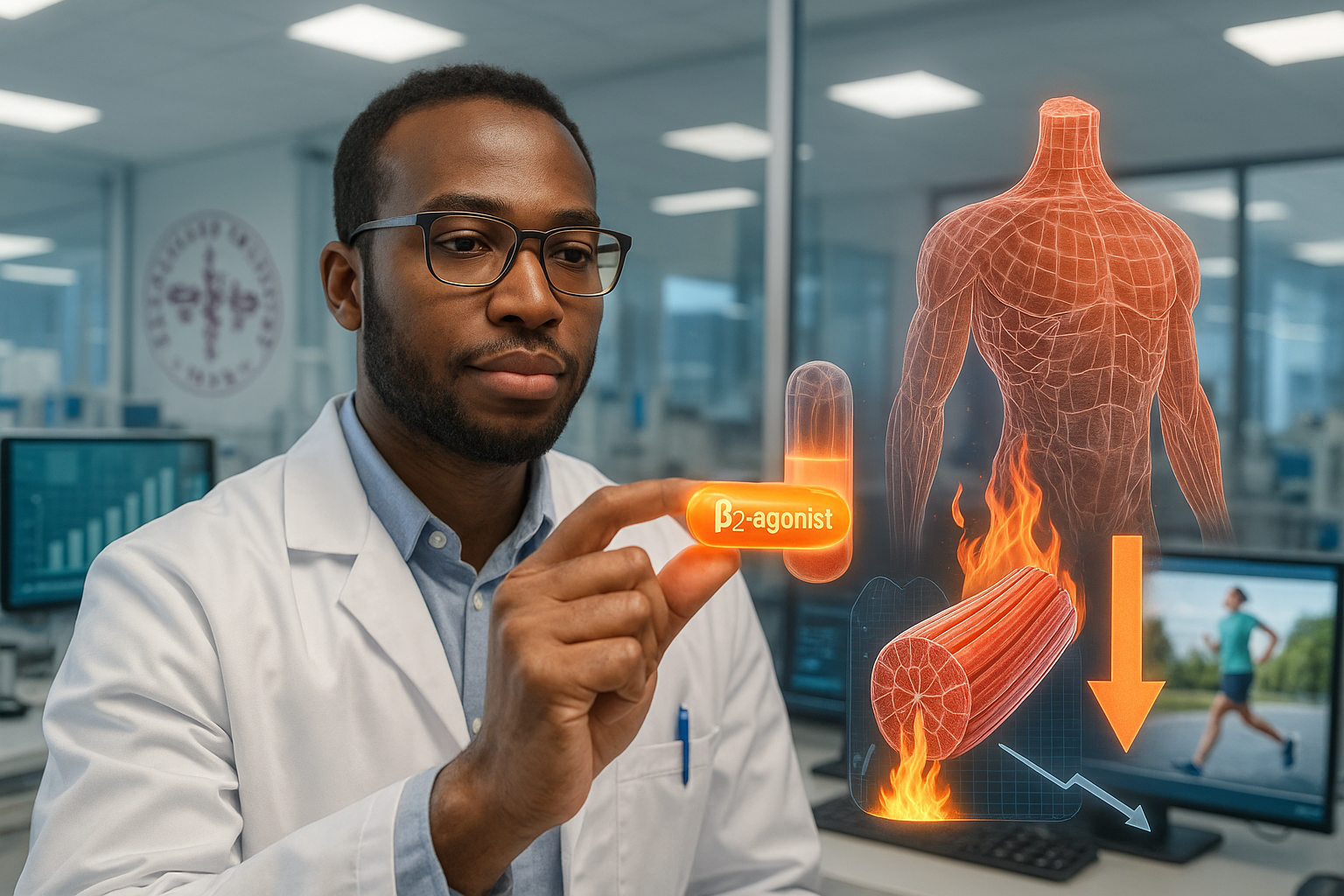
Researchers at Karolinska Institutet and Stockholm University have developed an experimental oral drug that boosts metabolism in skeletal muscle, improving blood sugar control and fat burning in early studies without reducing appetite or muscle mass. Unlike GLP-1-based drugs such as Ozempic, the candidate acts directly on muscle tissue and has shown good tolerability in an initial clinical trial, according to the study authors.
A study published in the journal Cell reports on a novel oral drug candidate for type 2 diabetes and obesity that primarily acts by increasing metabolic activity in skeletal muscle rather than suppressing appetite, according to researchers from Stockholm University and Karolinska Institutet. The work is summarized by Karolinska Institutet and Stockholm University and is based on the article “GRK-biased adrenergic agonists for the treatment of type 2 diabetes and obesity.”
The compound, taken in tablet form, has a different mechanism of action than GLP-1-based drugs such as Ozempic, which are administered as injections and act mainly through gut–brain signaling to reduce hunger. GLP-1 drugs are effective for blood sugar control and weight loss but often cause side effects including loss of appetite, gastrointestinal problems and, in some patients, reductions in lean or muscle mass, according to background material cited by the universities.
By contrast, the new substance targets skeletal muscle directly and activates its metabolism. In preclinical animal studies, it improved blood sugar control and body composition, with increased fat burning while preserving lean or muscle mass, according to summaries from Karolinska Institutet and Stockholm University. Muscle tissue plays a central role in whole-body glucose metabolism, and higher muscle mass is generally associated with better metabolic health and physical function, though the Cell paper does not quantify effects on life expectancy.
Early clinical data are limited but encouraging. An initial phase I, placebo‑controlled trial of the lead candidate enrolled 48 healthy volunteers and 25 people with type 2 diabetes and found that the oral tablet was well tolerated and showed pharmacokinetic properties consistent with once‑daily dosing, according to the Cell article and accompanying institutional releases. The trial was designed to assess safety and pharmacokinetics; efficacy in patients will be addressed in later‑stage studies.
“This drug represents a completely new type of treatment and has the potential to be of great importance for patients with type 2 diabetes and obesity. Our substance appears to promote healthy weight loss and, in addition, patients do not have to take injections,” said Shane C. Wright, assistant professor at Karolinska Institutet’s Department of Physiology and Pharmacology, in a statement published by Stockholm University.
The active ingredient is a newly developed β2-adrenergic receptor agonist engineered to preferentially engage GPCR kinase (GRK)–dependent signaling pathways in skeletal muscle. According to the Cell study, this GRK‑biased profile is intended to support glucose uptake and metabolic benefits in muscle while limiting the Gs/cAMP‑mediated cardiac stimulation and desensitization that have made traditional β2 agonists unattractive as chronic diabetes therapies.
Because it acts through a distinct mechanism from GLP-1 receptor agonists, the drug candidate may be effective either on its own or in combination with GLP‑1 medicines, the study authors note. In animal models, co‑treatment with a GLP‑1‑based therapy suggested the potential to counteract muscle loss sometimes observed with incretin‑based weight‑loss regimens, according to Karolinska Institutet.
Atrogi AB, the company developing the treatment, funded the clinical trial. The firm plans a larger phase II study to test whether the favorable effects seen in preclinical models and early human data translate into improved glucose control and body composition in people with type 2 diabetes or obesity, according to Stockholm University. Atrogi’s chief scientific officer and co‑founder, Professor Tore Bengtsson of Stockholm University’s Department of Molecular Biosciences, is a co‑author of the Cell paper and an inventor on related patent applications.
The research reflects an international collaboration involving scientists at Karolinska Institutet and Stockholm University, as well as Uppsala University, the University of Copenhagen, Monash University and the University of Queensland. Funding came from several sources, including the Swedish Research Council, the Swedish Society for Medical Research and the Novo Nordisk Foundation, among others, according to Stockholm University.
The authors and institutions emphasize that while the findings point to a possible future in which metabolic health can be improved without reducing appetite or muscle mass, the evidence in patients is still preliminary. Larger and longer clinical trials will be needed to determine how well the drug works in real‑world populations and how it compares with, or complements, existing GLP‑1‑based treatments.