Researchers from the University of Geneva and Lausanne University Hospital report they have visualized, in three dimensions and under near-native conditions, how cytotoxic T cells organize their killing machinery at the immune synapse. The work, published in Cell Reports, applies cryo-expansion microscopy to human T cells and to tumor tissue samples, providing nanoscale views intended to support immunology and cancer research.
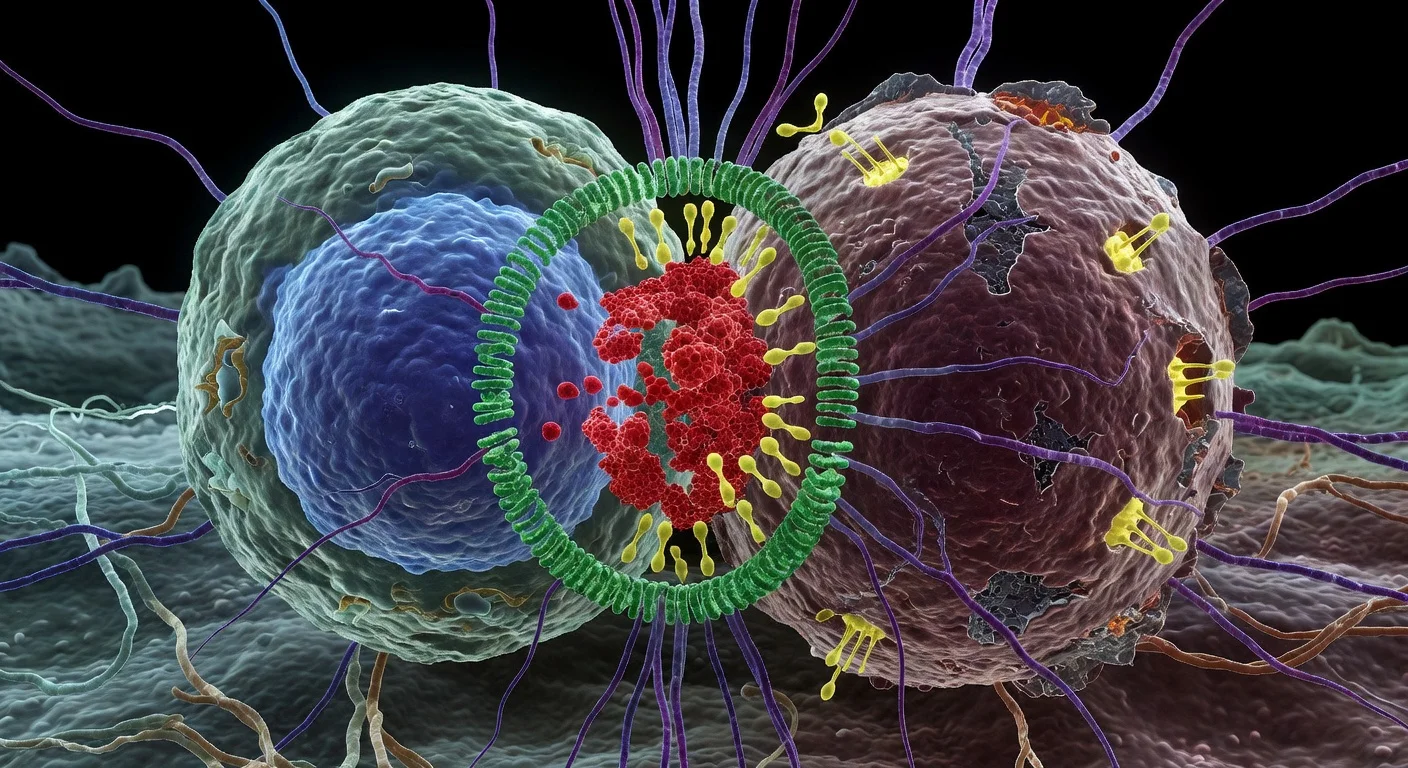
Cytotoxic T lymphocytes (CTLs) are immune cells that can eliminate infected or cancerous cells. Their killing activity is coordinated at a specialized contact site with the target cell known as the immune synapse, where CTLs deploy toxic molecules while limiting damage to neighboring cells.
In a Cell Reports study, scientists at the University of Geneva (UNIGE) and Lausanne University Hospital (CHUV) used cryo-expansion microscopy (cryo-ExM)—a method that rapidly freezes samples into a vitreous state and then physically expands them in a hydrogel—to examine the three-dimensional organization of CTLs and immune synapses in a near-native state.
The researchers report that the contact zone can form a dome-like membrane architecture, and they describe structural variation in cytotoxic granules, including granules with single or multiple dense “cores” that concentrate killing molecules.
Beyond experiments in cultured cells, the team says it adapted the approach to human tumor tissue, enabling visualization of tumor-infiltrating T cells and their cytotoxic machinery at nanometer-scale detail in a clinical-context sample.
The authors say the imaging framework could help researchers better analyze how immune-cell structure relates to function, including questions relevant to immuno-oncology.