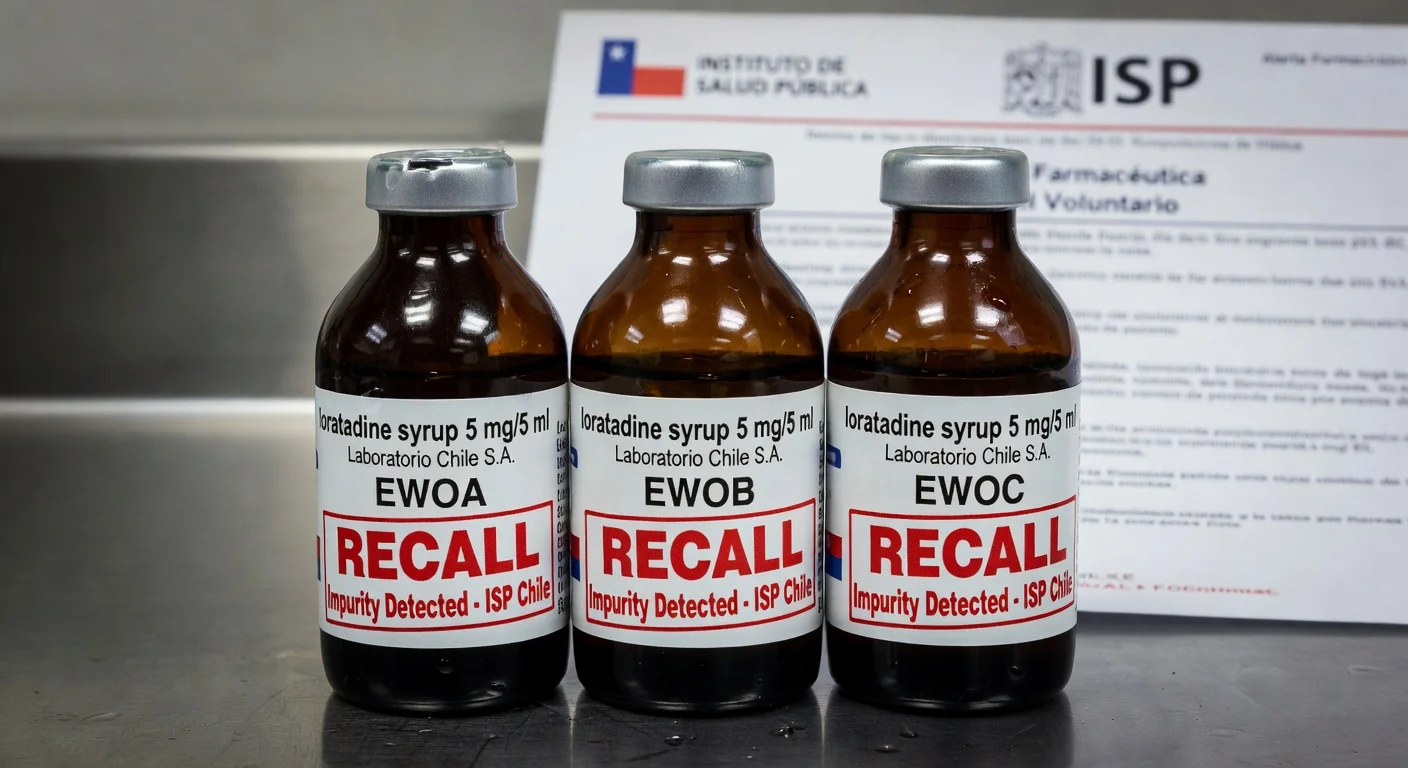
Chile's Public Health Institute (ISP) issued a pharmaceutical alert ordering the voluntary recall of Laboratorio Chile S.A.'s loratadine syrup 5 mg/5 ml after detecting out-of-specification impurities in stability studies. The action targets batches EWOA, EWOB, and EWOC, expiring in September 2026.
Chile's Public Health Institute (ISP) issued alert N°09/26 on Monday, classifying the recall as voluntary and initiated by the sanitary registration holder, Laboratorio Chile S.A. The product, a 60 ml bottle with registration F-8414, is an antihistamine used to treat allergic symptoms like rhinitis and urticaria.
The affected batches are EWOA, EWOB, and EWOC, identified in an ongoing stability study showing impurities exceeding required specifications. These batches could still be in circulation, with expiration in September 2026.
ISP activated protocols to identify, trace, and remove the batches from pharmacies, health centers, and distributors, notifying professionals and establishments. The voluntary recall aims to prevent risks as a precautionary measure under sanitary supervision.
No adverse events linked to the product have been reported, per the alert.