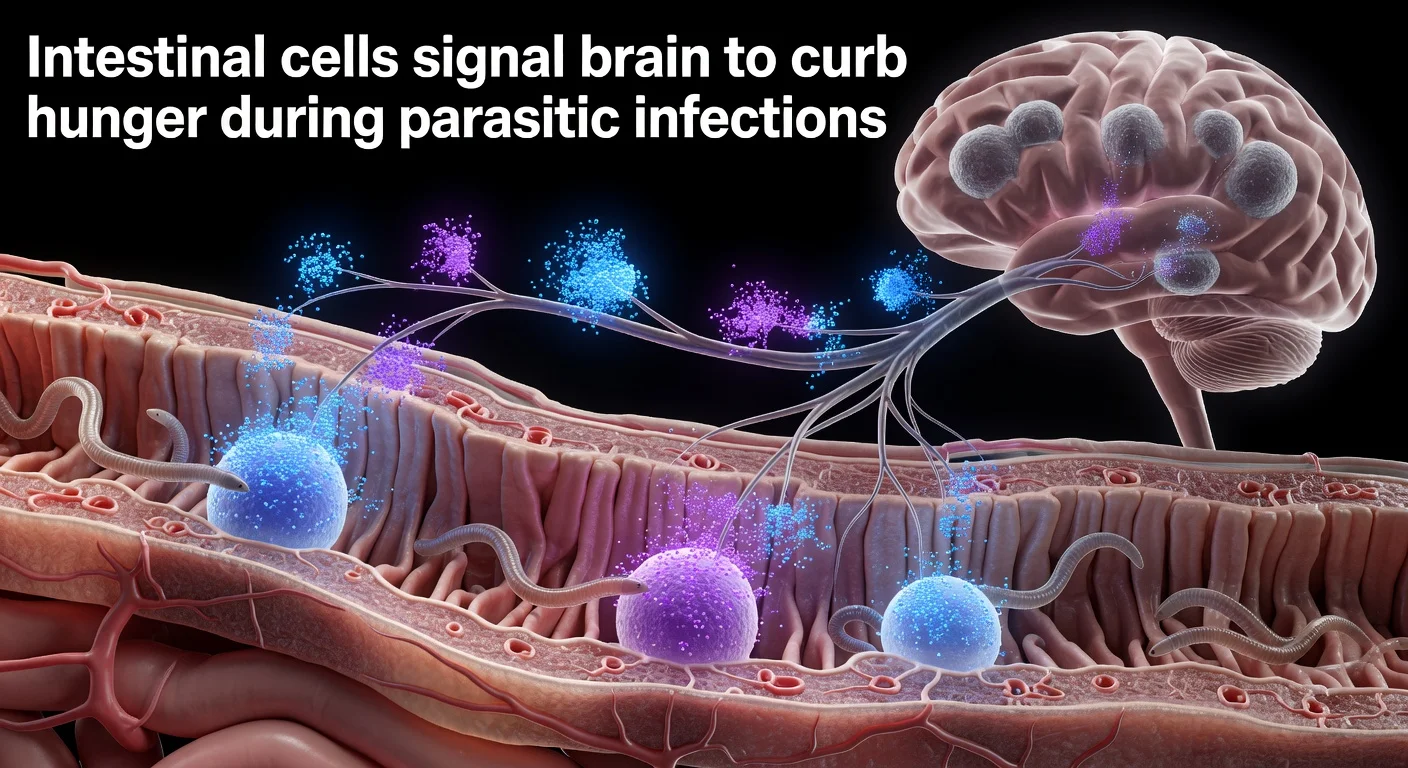
A team led by David Julius, the 2021 Nobel Prize winner in Medicine, has described the molecular mechanism by which intestinal tuft cells signal the brain to suppress appetite during parasitic infections. Published today in Nature, the study identifies communication via acetylcholine and serotonin that activates the vagus nerve. The finding could aid treatments for conditions like irritable bowel syndrome.
David Julius's team at the University of California, San Francisco (UCSF), has traced the pathway linking the intestinal immune system to the brain during parasitic worm infections. Tuft cells detect parasites via succinate and release acetylcholine in two phases: an initial brief burst and a sustained release after full immune response. This acetylcholine stimulates enterochromaffin (EC) cells, which release serotonin and activate vagus nerve fibers to the brain, causing appetite loss akin to gastroenteritis, according to the study published Wednesday in Nature. First author Koki Tohara explained: “Tuft cells do something neurons do, but through a completely different mechanism,” using acetylcholine without typical neuronal machinery. Julius noted: “The gut waits to confirm the threat is real and persistent before telling the brain to change your behavior,” explaining delayed symptoms. Coauthor and UCSF immunologist Richard Locksley highlighted interest in how these non-synaptically connected cells alter behavior. Mouse experiments confirmed the mechanism: animals with intact tuft cells ate less, while genetically modified ones lacking acetylcholine production maintained normal intake. The authors suggest applications for managing infection responses and conditions like food intolerances or chronic visceral pain, as tuft cells exist in other epithelia like airways. Spanish experts such as Félix Viana from the Alicante Institute of Neurosciences call it “interesting” and relevant for defenses in other tissues, while José Luis Trejo from the Cajal Neuroscience Center views it as a “sensory interface” with therapeutic potential.