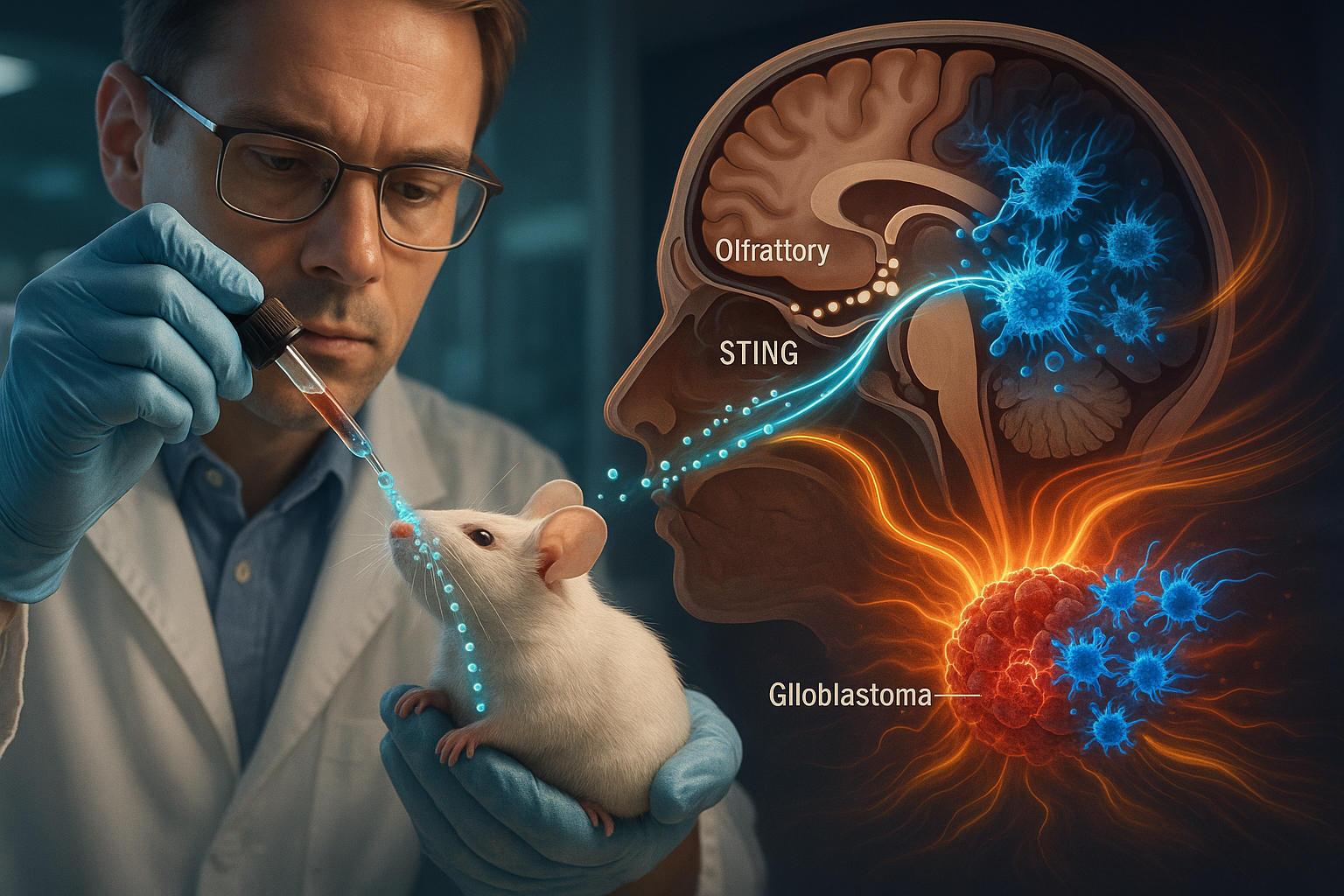
Researchers at Washington University School of Medicine in St. Louis, working with scientists at Northwestern University, have developed a noninvasive nasal nanotherapy that activates the immune system to attack aggressive brain tumors in mice. By delivering spherical nucleic acids that trigger the STING immune pathway directly from the nose to the brain, the approach eliminated glioblastoma tumors in mouse models when combined with drugs that boost T-cell activity, according to a study in the Proceedings of the National Academy of Sciences.
Glioblastoma, the most common malignant brain tumor, affects about three in every 100,000 people in the United States and progresses rapidly, with the disease almost always proving fatal. It develops from astrocytes, a type of brain cell, and is particularly hard to treat because it is difficult to deliver drugs across the blood-brain barrier.
In a new preclinical study, researchers at Washington University School of Medicine in St. Louis and Northwestern University report a promising alternative: spherical nucleic acids built on gold nanoparticle cores that activate the STING immune pathway when delivered as nasal drops. The work, led by co-corresponding authors Alexander H. Stegh, PhD, of Washington University, and Chad A. Mirkin, PhD, of Northwestern University, is described in the journal Proceedings of the National Academy of Sciences.
The STING pathway — short for stimulator of interferon genes — helps trigger immune defenses when cells detect foreign DNA, and has been a major target for turning "cold" tumors such as glioblastoma into "hot" tumors that respond to immunotherapy. Past studies have shown that drugs designed to activate STING can prime the immune system to attack glioblastoma, but these agents break down quickly in the body and typically must be injected directly into the tumor, often requiring repeated, highly invasive procedures.
"We wanted to change this reality and develop a noninvasive treatment that activates the immune response to attack glioblastoma," Stegh said, according to Washington University. He added that the team’s results show that precisely engineered nanostructures known as spherical nucleic acids can safely and effectively activate powerful immune pathways within the brain.
To address the limitations of existing STING agonists, the researchers worked with Mirkin, who invented spherical nucleic acids — nanoscale structures in which DNA or RNA strands are densely arranged around a nanoparticle core. Together, the teams designed a new class of spherical nucleic acids featuring gold nanoparticle cores coated with short DNA fragments that activate the STING pathway in specific immune cells. For delivery to the brain, they chose the nose as the entry route.
Intranasal therapy has been explored as a way to target medications to the brain, but, according to Washington University and ScienceDaily, no nanoscale therapy had previously demonstrated the ability to activate immune responses against brain cancers via this route. "We really wanted to minimize patients having to go through [direct tumor injections] when they are already ill, and I thought that we could use the spherical nucleic acid platforms to deliver these drugs in a noninvasive way," said first author Akanksha Mahajan, PhD, a postdoctoral research associate in Stegh’s lab.
The team tagged the spherical nucleic acids with a molecular label visible under near-infrared light to track their movement. In mice with glioblastoma, nanodrops administered into the nasal passages traveled along the path of the main nerve that connects facial regions to the brain. Once in the brain, the nanomedicine concentrated in immune cells within and around the tumor and also showed activity in nearby lymph nodes, while largely avoiding distribution to other organs, the researchers report.
Analyses of immune cells in and near the tumors showed that the therapy activated the cGAS–STING pathway and created a more proinflammatory tumor microenvironment enriched with effector T cells and proinflammatory macrophages, consistent with the underlying PNAS study.
When the intranasal nanotherapy was combined with drugs designed to activate T lymphocytes — such as immune checkpoint inhibitors — one or two doses eradicated glioblastoma tumors in mouse models and induced long-term immunity that prevented the cancers from returning, according to Washington University and the PNAS report. The combined treatment inhibited tumor growth more effectively and produced longer survival than currently tested STING-targeting therapies in similar models.
Stegh cautioned that simply switching on the STING pathway is unlikely to cure glioblastoma by itself because the tumor uses multiple mechanisms to blunt or shut down immune responses. His group is now exploring ways to add further immune-activating capabilities to the nanostructures so that several therapeutic targets can be addressed in a single treatment.
The study, published in the November 2025 issue of Proceedings of the National Academy of Sciences, was supported by grants from the National Cancer Institute and other National Institutes of Health programs, as well as by disease-focused foundations and institutional cancer center funding, according to Washington University and ScienceDaily. Disclosures include Stegh’s shareholding in Exicure Inc., which develops spherical nucleic acid therapeutic platforms, and Mirkin’s shareholding in Flashpoint Therapeutics, which develops SNA-based therapeutics. Stegh and Mirkin are also co-inventors on a patent describing SNA nanoconjugates designed to cross the blood-brain barrier.