Researchers at the Institut Pasteur and Inserm have developed a triple-drug strategy that induces necroptosis in malignant B cells, triggering a strong anti-tumor immune response in preclinical models of leukemia. By reprogramming how cancer cells die, the approach enabled complete leukemia elimination in animals and may offer a new avenue for treating B cell-related blood cancers, according to findings published in Science Advances.
In a preclinical advance for cancer immunotherapy, scientists at the Institut Pasteur and Inserm have shown that a combination of three existing drugs can force malignant B cells to undergo necroptosis, a form of programmed cell death that emits danger signals and activates the immune system. The work, described by the team and summarized by Institut Pasteur and ScienceDaily, suggests that manipulating the way tumor cells die can substantially strengthen anti-tumor immunity.
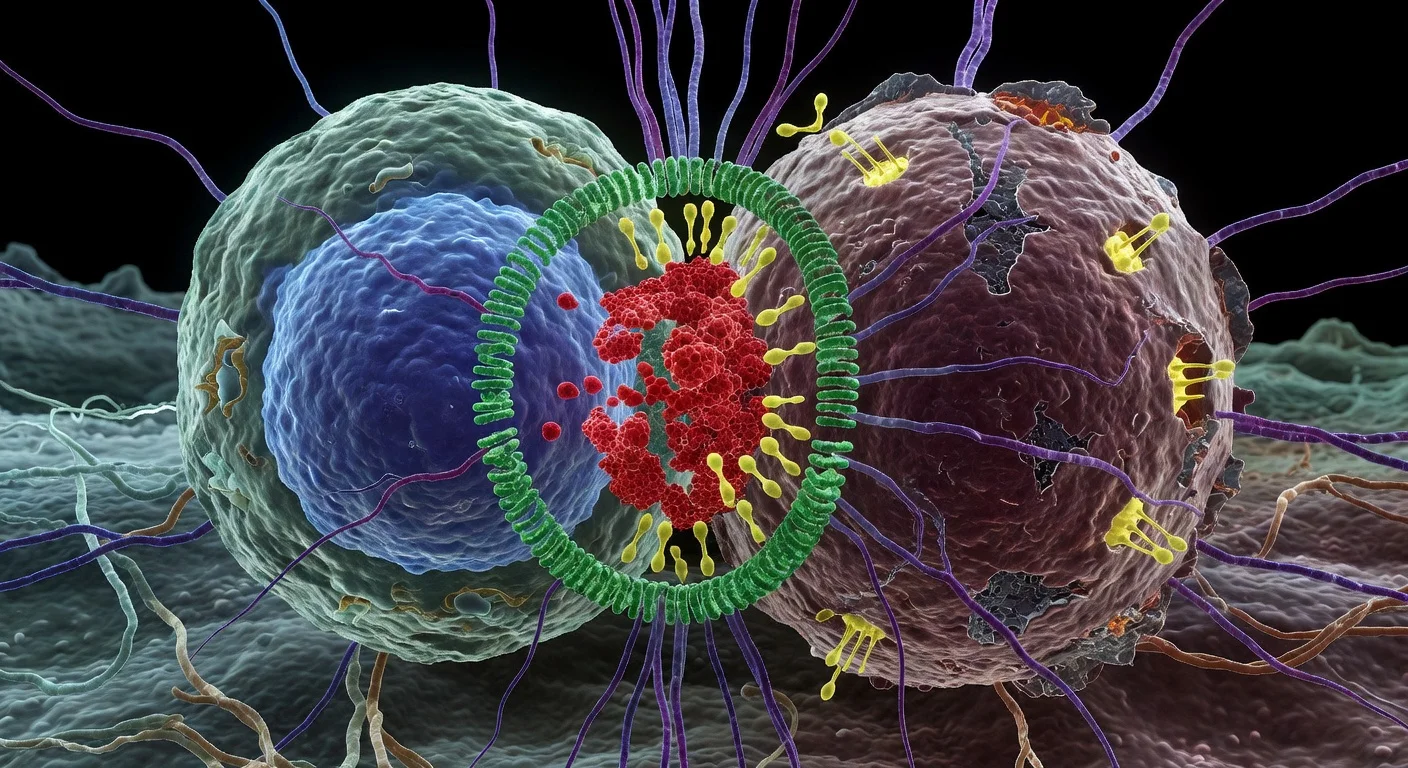
Immunotherapy aims to help a patient's own immune cells seek out and destroy tumor cells. The researchers focused on blood cancers involving B cells, such as certain leukemias and lymphomas. Their initial experiments showed that malignant B cells are generally resistant to necroptosis because they lack sufficient levels of MLKL, a protein essential for this pathway.
To overcome this barrier, the team devised a triple-drug regimen using agents already approved for clinical use. According to the Institut Pasteur press release and coverage in ScienceDaily, this combination reprogrammed malignant B cells to die through necroptosis and released strong immune-stimulating signals. In preclinical mouse models, the strategy led to complete elimination of leukemia, indicating potent immune-mediated tumor control.
"The triple therapy we used forces cancer cells to die in a way that activates the immune system," said Philippe Bousso, Inserm Research Director and Head of the Institut Pasteur's Dynamics of Immune Responses Unit, in statements quoted by Institut Pasteur and ScienceDaily.
To understand how different forms of cell death shape immune activity, the researchers used advanced intravital imaging. This real-time imaging technique allowed them to visualize immune cells interacting with dying cancer cells in living animals and to compare how necroptosis versus other death mechanisms influenced immune behavior.
"This novel immunotherapy strategy, successfully tested in preclinical models, turns tumor cells into triggers for the immune system, pointing to a potential therapeutic avenue for certain cancers, such as lymphomas or leukemias affecting B cells," Bousso explained in comments reported by Institut Pasteur. He added, "By changing the way cancer cells die, we can harness the support of our immune system to fight against the tumor."
The study, led by first author Ruby Alonso and colleagues in the Dynamics of Immune Responses Unit, was published on August 15, 2025, in Science Advances (volume 11, issue 33) under the title "Reprogramming RIPK3-induced cell death in malignant B cells promotes immune-mediated tumor control." The work was supported by several institutions, including the European Research Council and the ARC Foundation for Cancer Research, as noted by Institut Pasteur.