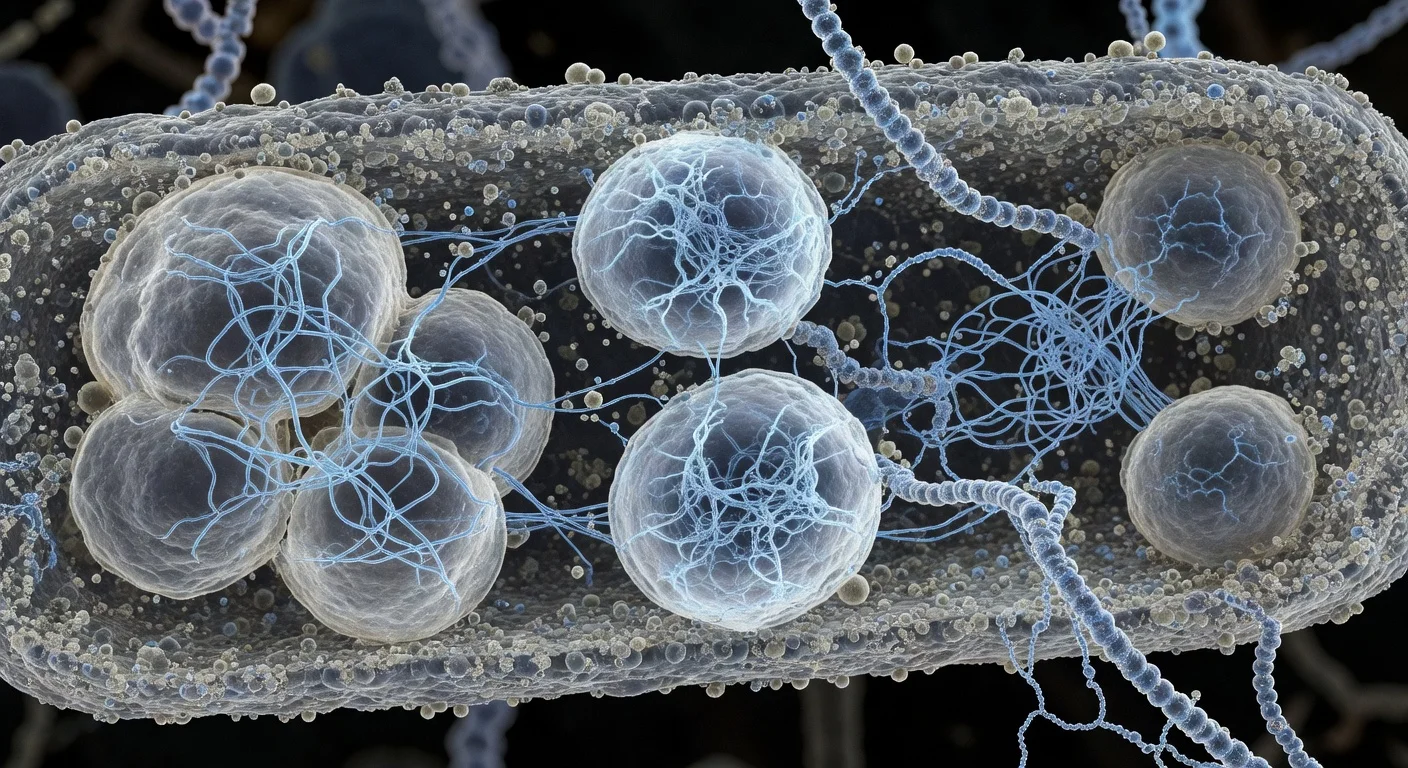
Scientists at Scripps Research report that some biomolecular condensates—membrane-less, droplet-like cellular compartments—contain networks of thin protein filaments that act as an internal scaffold. The team says disrupting this filament architecture alters condensate physical properties and impairs bacterial growth and DNA segregation, raising the possibility that condensate structure could one day be therapeutically targetable in diseases such as cancer and ALS. The study appeared in Nature Structural & Molecular Biology on February 2, 2026.
Biomolecular condensates are droplet-like clusters that help cells organize key activities without membranes. Researchers describe them as involved in processes that include regulating how genetic instructions in DNA are turned into proteins, helping clear potentially toxic cellular waste, and contributing to mechanisms that can suppress tumor growth.
In work focused on a bacterial protein called PopZ, a Scripps Research-led team examined how these membrane-less compartments can have functional organization. In certain rod-shaped bacteria, PopZ accumulates at the cell poles and forms condensates that recruit other proteins needed for cell-division-related processes.
Using cryo-electron tomography—an approach the researchers liken to a CT scan at molecular scale—the team reports that PopZ molecules assemble into thin filaments through an ordered, step-by-step process. Those filaments form an internal scaffold that helps determine the condensate’s physical characteristics.
The study also used single-molecule Förster resonance energy transfer (FRET) to probe PopZ behavior at the level of individual molecules. The researchers report that PopZ adopts different conformations depending on whether it is inside or outside the condensate. “Realizing that protein conformation depends on location gives us multiple ways to engineer cellular function,” said Daniel Scholl, the paper’s first author and a former postdoctoral researcher in the Lasker and Deniz labs.
To test whether the filament network is required for normal function, the team engineered a PopZ variant that could not form filaments. According to the researchers, the altered condensates were more fluid and had reduced surface tension. When introduced into bacteria, the changes were associated with halted growth and failures in DNA segregation.
Although the experiments centered on a bacterial system, Scripps Research said the findings may inform how scientists think about condensates in human cells as well. The research organization pointed to filament-based condensates implicated in protein quality control and growth regulation—processes tied to neurodegenerative disease and cancer biology—and suggested that definable condensate architecture could eventually provide new therapeutic entry points.
The paper, titled “The filamentous ultrastructure of the PopZ condensate is required for its cellular function,” lists Keren Lasker as senior author, with Ashok A. Deniz and Raphael Park as co-corresponding authors. Additional authors named in the research organization’s release include Tumara Boyd, Andrew P. Latham, Alexandra Salazar, Asma M. A. M. Khan, Steven Boeynaems, Alex S. Holehouse, Gabriel C. Lander and Andrej Sali.
Scripps Research said the work was supported by funders including the National Institutes of Health and the National Science Foundation, among others.