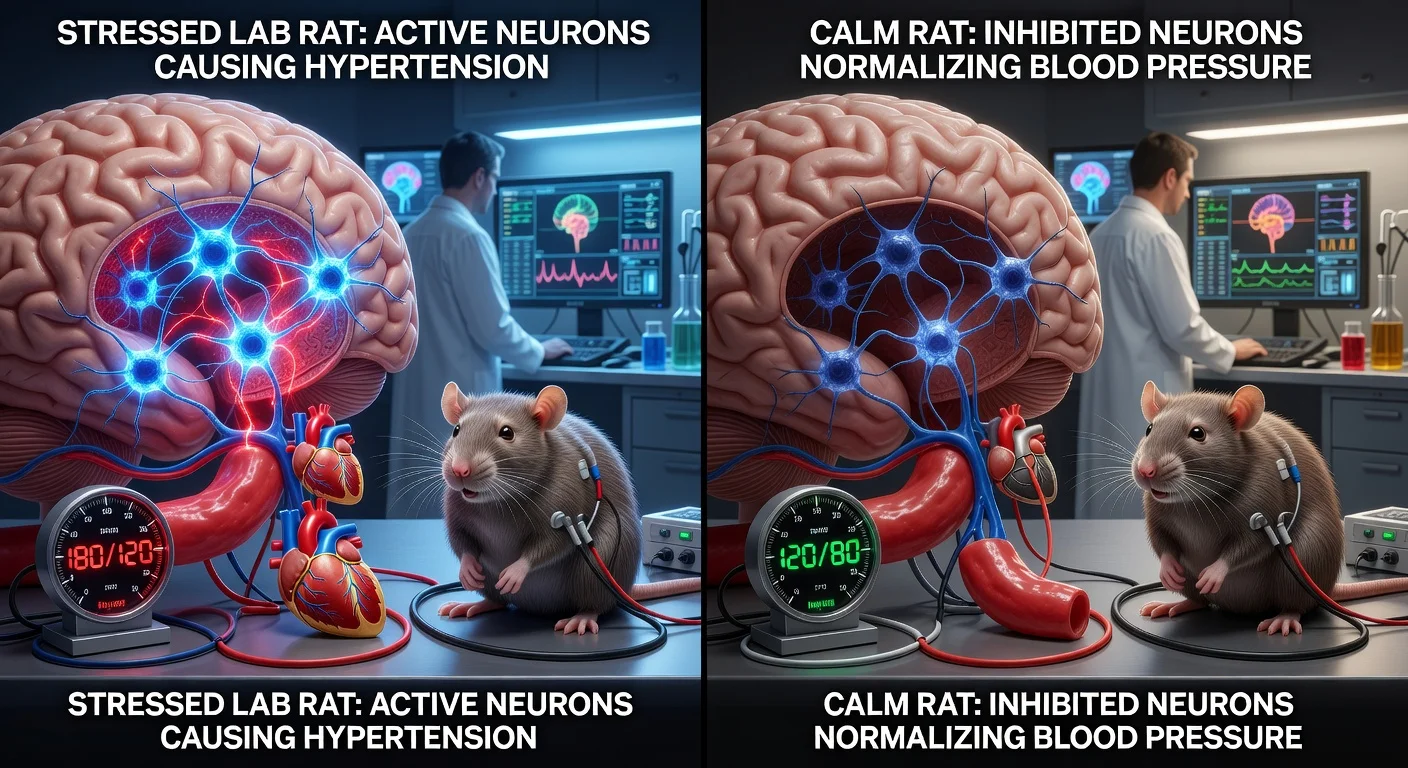
Researchers working with the University of Auckland and Brazil’s University of São Paulo report that neurons in the medulla’s lateral parafacial (pFL) region—best known for helping drive forceful exhalations—also amplify sympathetic nerve activity and can raise blood pressure in an animal model of neurogenic hypertension. Inhibiting these neurons lowered blood pressure toward normal in hypertensive rats, findings published in Circulation Research.
A research team including Professor Julian Paton of the University of Auckland’s Manaaki Manawa – Centre for Heart Research and collaborators in Brazil has reported evidence that a small brainstem region involved in breathing may contribute to high blood pressure in an animal model.
According to the University of Auckland’s release, the lateral parafacial region in the brainstem is recruited during forceful exhalations—such as laughing, coughing or exercise—which use abdominal muscles rather than relying primarily on the lungs’ elastic recoil. Paton said the region is “recruited into action” during these “forced” exhalations.
In the peer-reviewed paper, the researchers focused on neurons in the medullary lateral parafacial (pFL) region and tested how they influence sympathetic pathways that regulate blood vessel tone. In experiments in rats, optogenetic activation of pFL neurons triggered active expiration and increased sympathetic activity during expiration, raising arterial pressure. The study also reported that pFL neurons project to presympathetic neurons in the rostral ventrolateral medulla (RVLM) and the pontine A5 region; in hypertensive animals, synaptic transmission in the pFL-to-RVLM pathway was enhanced.
To model neurogenic hypertension, the researchers used chronic intermittent hypoxia in rats. They reported that pharmacogenetic inhibition of pFL neurons eliminated expiratory-linked sympathetic excitation and normalized arterial pressure in hypertensive rats.
The University of Auckland release further argues that the pFL region can be driven by signals originating outside the brain—specifically from the carotid bodies, oxygen-sensing structures near the carotid artery in the neck. On that basis, Paton said the group aims to reduce carotid body activity with a repurposed drug to dampen the brainstem circuit “remotely,” potentially avoiding medications that must penetrate the brain.
The release also suggests the findings could be relevant to high blood pressure associated with sleep apnoea, where carotid body activity can rise when breathing pauses during sleep. The researchers emphasized the work was conducted in animals.
The study is titled “Lateral Parafacial Neurons Evoked Expiratory Oscillations Driving Neurogenic Hypertension,” published online December 17, 2025, and listed in the January 16, 2026 issue of Circulation Research (vol. 138, no. 2) with DOI: 10.1161/CIRCRESAHA.125.326674.