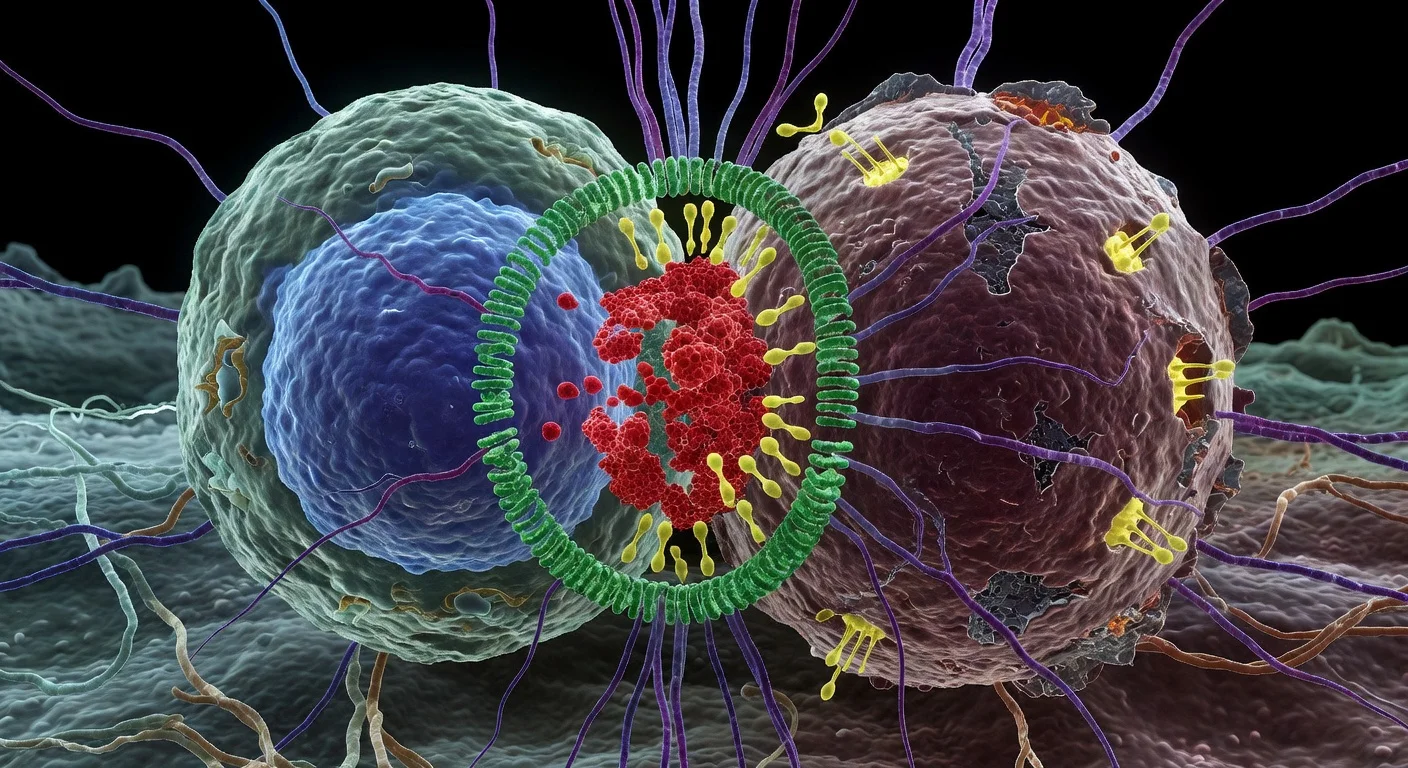
Researchers report that a single injection of a modified herpes virus draws immune cells deep into glioblastoma tumors, leading to longer survival in a clinical trial. The therapy, tested on 41 patients with recurrent brain cancer, activates T cells that persist and attack cancer cells. Findings were published in Cell.
Scientists from Mass General Brigham and Dana-Farber Cancer Institute have developed an oncolytic virus therapy that targets glioblastoma, the most aggressive form of primary brain cancer. The virus, a genetically engineered herpes simplex virus created by E. Antonio Chiocca, MD, PhD, replicates only in cancer cells, killing them and alerting the immune system without harming healthy tissue. In a phase 1 clinical trial with 41 patients suffering from recurrent glioblastoma, the single-dose treatment was linked to improved survival compared to historical data, particularly among those with pre-existing antibodies to the virus. Analysis of tumor samples revealed sustained infiltration by cytotoxic T cells, positioned near dying tumor cells in patients who lived longer post-treatment. The therapy also amplified existing T cells in the brain. Co-senior author Kai Wucherpfennig, MD, PhD, noted, 'Patients with glioblastoma have not benefited from immunotherapies that have transformed patient care in other cancer types such as melanoma because glioblastoma is a 'cold' tumor with poor infiltration by cancer-fighting immune cells. Findings from our clinical trial and our mechanistic study show that is now feasible to bring these critical immune cells into glioblastoma.' Chiocca added, 'We show that increased infiltration of T cells that are attacking tumor cells translates into a therapeutic benefit for patients with glioblastoma. Our findings could have important implications for a cancer whose standard of care hasn't changed for 20 years.' The study appears in Cell (2026; 189(5):1287).