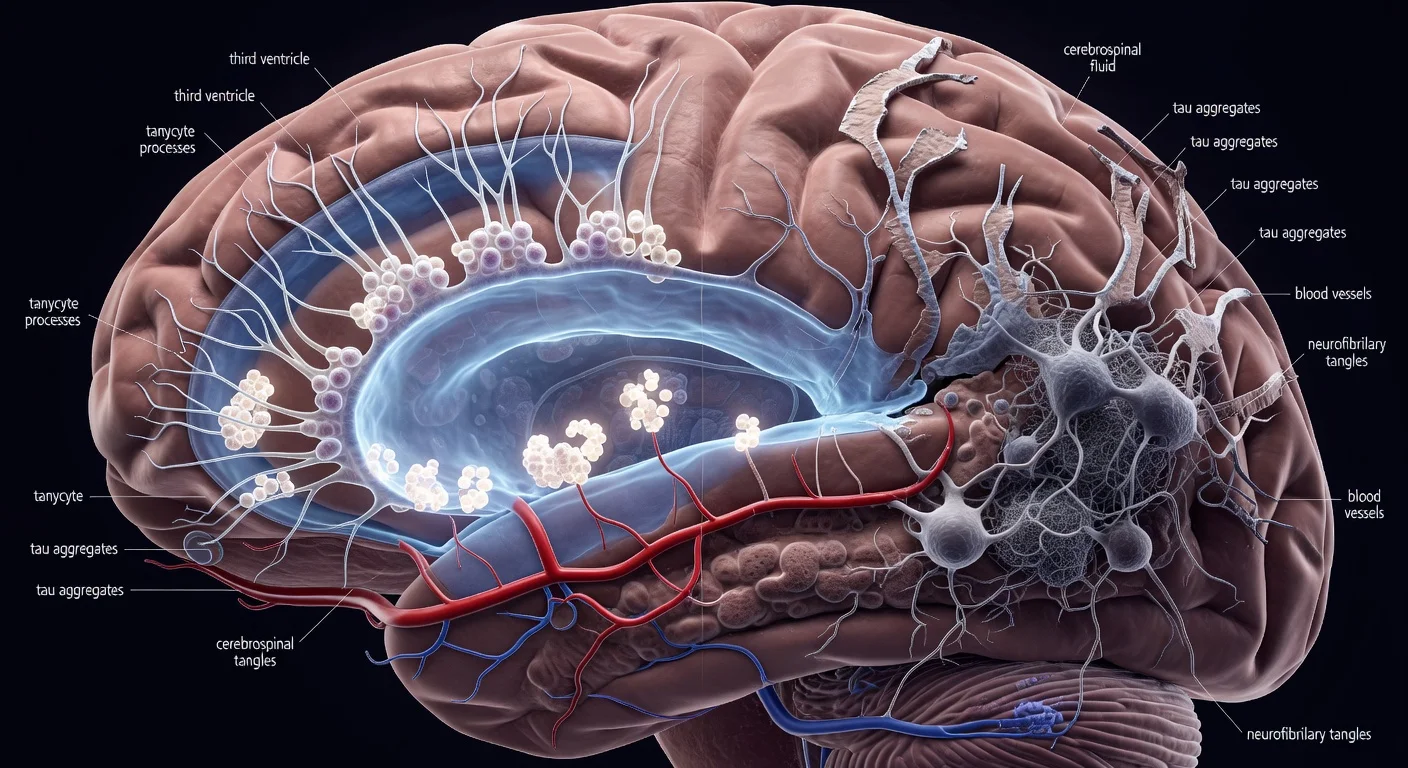
Researchers report that tanycytes—specialized cells lining the brain’s third ventricle—can help move tau protein from cerebrospinal fluid into the bloodstream, and that signs of tanycyte disruption in Alzheimer’s patient tissue may be associated with impaired tau removal. The findings, published March 5 in Cell Press Blue, are based on animal and cell experiments and analyses of human brain samples.
The buildup of tau protein in the brain is widely recognized as one of the hallmarks of Alzheimer’s disease. A study published March 5 in the Cell Press journal Cell Press Blue describes a mechanism involving tanycytes—specialized non-neuronal cells—that could contribute to how tau accumulates when clearance pathways fail.
Tanycytes are primarily located in the third ventricle. Prior work has linked them to shuttling metabolic signals between the blood and cerebrospinal fluid (CSF), the fluid that surrounds the brain and spinal cord and helps maintain internal balance.
In the new research, scientists used animal and cellular models alongside analyses of tissue from Alzheimer’s patients to examine whether tanycytes help clear harmful molecules such as tau. The study reports that tanycytes can transport tau from the CSF into the bloodstream, where it can be cleared from the body. When tanycytes are damaged or do not function properly, the researchers report that tau can accumulate.
“Surprisingly, we were able to show in rodent and cellular models not only that tanycytes were indeed involved in clearing tau but also that tanycytes in the brains of human Alzheimer’s patients were fragmented and had changes in gene expression related to this shuttle function,” said Vincent Prévot of INSERM in France, the study’s corresponding author.
Prévot added that the findings point to a potential, disease-relevant role for tanycytes in neurodegeneration, and suggested that protecting tanycyte function could be a possible strategy to improve tau clearance—though he and colleagues stressed that translating the results into therapies would require additional work.
The researchers also highlighted limitations that could affect how the results are interpreted, including the lack of animal models that fully replicate Alzheimer’s disease and the need for larger patient cohorts and more longitudinal data to better assess causality and the sequence of events linking tanycyte dysfunction with tau pathology.
The work was supported by the European Research Council, the U.S. National Institutes of Health, the Fondation pour la Recherche Médicale, and the Fondation NRJ for Neuroscience–Institut de France. The study is titled “Tanycytic degeneration impairs tau clearance and contributes to Alzheimer’s disease pathology” (DOI: 10.1016/j.cpblue.2026.100003).