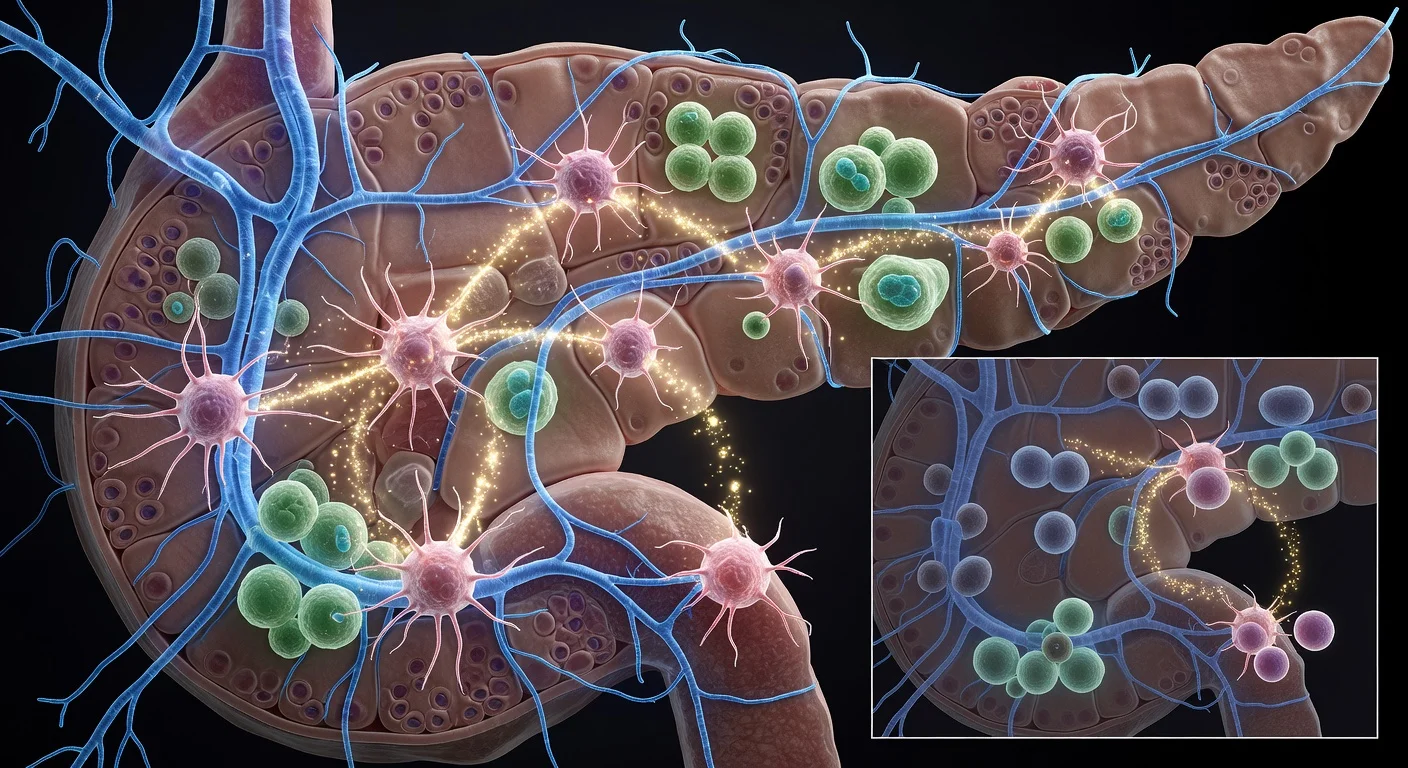
Researchers at Cold Spring Harbor Laboratory report that support cells known as myofibroblastic cancer-associated fibroblasts (myCAFs) can recruit sympathetic nerve fibers into early pancreatic lesions, creating a feedback loop that may help pancreatic cancer take hold before full tumors form. In mouse experiments, disrupting sympathetic nerve activity reduced fibroblast activation and was associated with nearly a 50% reduction in tumor growth.
Pancreatic cancer is difficult to detect early and often responds poorly to standard treatments, factors that have driven scientists to look for new ways to slow the disease before it advances.
A team at Cold Spring Harbor Laboratory (CSHL), led by postdoctoral researcher Jérémy Nigri in the laboratory of Professor David Tuveson, focused on how the nervous system may influence the earliest stages of pancreatic cancer. Nigri noted that researchers have long recognized perineural invasion—a process in which cancer cells spread along nerves—as one way pancreatic tumors can disseminate.
Nerves present before tumors fully form
In the new work, the researchers report evidence that nerves may play an active role even earlier than perineural invasion, appearing in and around pre-cancerous pancreatic lesions.
Using whole-mount immunofluorescence to generate three-dimensional images, the team observed dense nerve networks interwoven through pancreatic lesions and closely associated with tumor-promoting fibroblasts known as myCAFs. In conventional two-dimensional views, the same nerve structures can appear as small, scattered puncta.
“When we first saw this picture, I was shocked,” Nigri said. “I couldn’t even imagine the lesion like this. I’d only ever seen it in 2D.”
A proposed feedback loop involving sympathetic nerves
Based on experiments in mice and human cell systems, the researchers describe a cycle in which myCAFs emit signals that attract fibers from the sympathetic nervous system, which governs the body’s fight-or-flight response. Those nerve fibers release norepinephrine, and the study reports that norepinephrine can bind to fibroblasts and trigger a rise in intracellular calcium. The resulting calcium surge further activates myCAFs, a response the researchers say can promote pre-cancerous growth while drawing in additional nerve fibers—reinforcing the loop.
Interrupting nerve activity reduced tumor growth in mice
To test whether disrupting this signaling could slow disease, the team used a neurotoxin in one mouse experiment to disable sympathetic nervous system activity. They reported reduced fibroblast activation and a nearly 50% reduction in tumor growth.
“In one experiment, we use a neurotoxin to disable the sympathetic nervous system,” Nigri said. “We show reduced fibroblast activation and a nearly 50% reduction in tumor growth.”
Treatment implications and next steps
The findings were published in Cancer Discovery, a journal of the American Association for Cancer Research. The researchers suggest that because the nerve–myCAF interaction appears early, it may represent a potential therapeutic target. They also point to clinically available drugs—including doxazosin—as candidates to explore in combination with established approaches such as chemotherapy or immunotherapy.
“The next step will be to study this more in detail and try to find a way to block the crosstalk between fibroblasts and nerves,” Nigri said.
CSHL said the work was supported by multiple funders, including the Lustgarten Foundation and the Pancreatic Cancer Action Network.