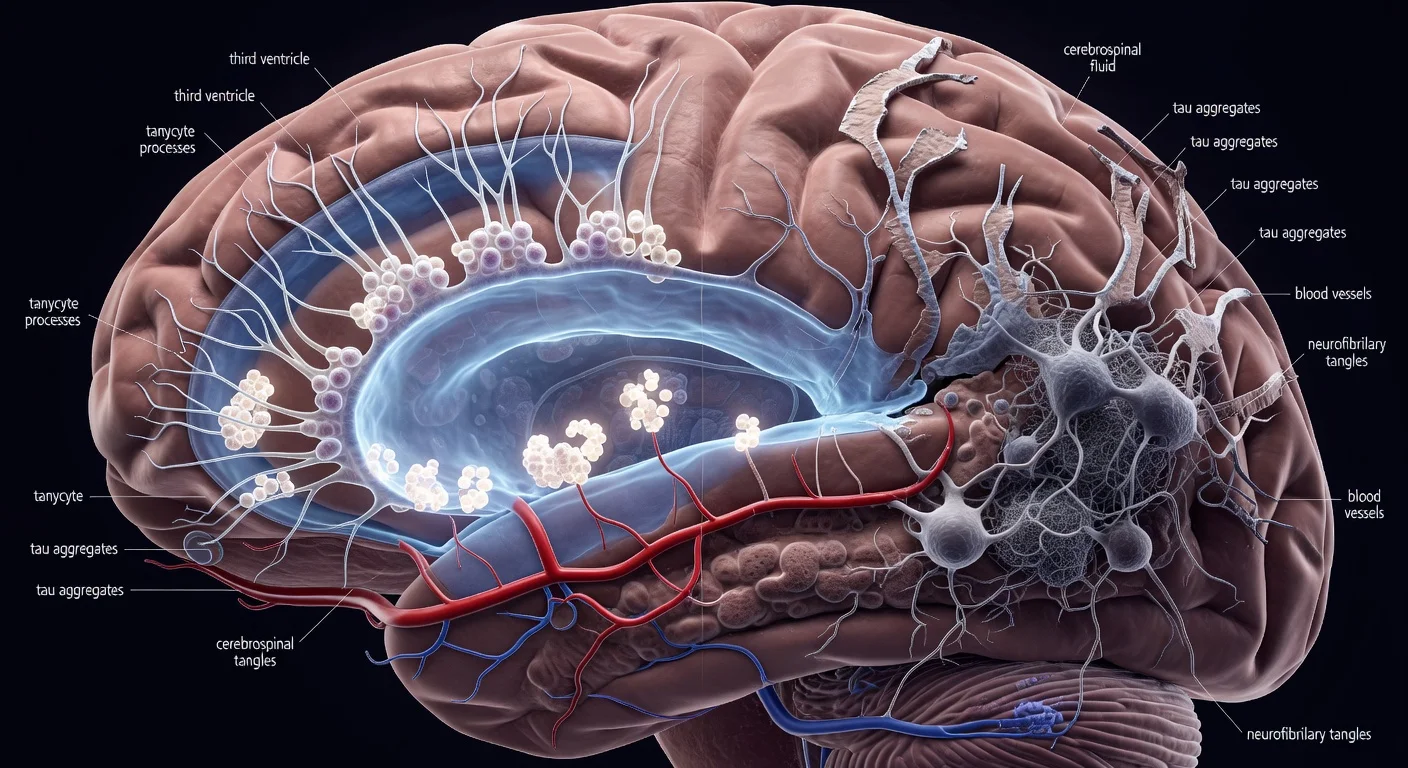
Researchers have found that repeated head impacts in contact sports damage the blood-brain barrier, potentially driving chronic traumatic encephalopathy (CTE) in former athletes. The discovery, based on MRI scans of retired footballers, rugby players and boxers, suggests new diagnostic and preventive approaches. Drugs to strengthen the barrier may help avert the condition.
A study published in Science Translational Medicine reveals that long-term damage to the blood-brain barrier from repetitive head knocks contributes to chronic traumatic encephalopathy (CTE), a neurodegenerative condition affecting some former footballers, rugby players and boxers. CTE, which causes cognitive issues, memory problems, depression and emotional instability, is currently only diagnosable post-mortem via tau protein buildup in autopsies. DOI: 10.1126/scitranslmed.adu6037. Matthew Campbell at Trinity College Dublin and colleagues scanned the brains of 47 retired contact-sport athletes, averaging 12 years post-retirement, using an MRI contrast agent that detects barrier breaches. In 17 participants, the agent leaked extensively into brain tissue, unlike in non-contact athletes like rowers or non-athletes. Those with greater leakage scored worse on cognitive and memory tests, indicating barrier damage as an early CTE factor. Chris Greene at the Royal College of Surgeons in Ireland explained that impacts loosen cell seals in the barrier, a dynamic system of tightly packed cells lining brain blood vessels. This allows blood proteins, immune cells and inflammatory substances to enter, causing inflammation and exacerbating tau misfolding from head blows. Post-mortem CTE brains showed immune infiltration, mirroring Alzheimer's features. Michael Buckland at the University of Sydney noted this strengthens prior evidence linking barrier disruption to CTE. The MRI method could enable living diagnoses for symptomatic individuals and monitor at-risk athletes, pending further research. Greene suggested repurposing drugs like bevacizumab to reduce vessel leakiness or minocycline for inflammation, intervening early before tau pathology entrenches. Campbell added, “There are many drugs in development that are seeking to restore the blood-brain barrier for the treatment of neurological disorders, so the future will be very bright if we can see the approval of some of these medications.”