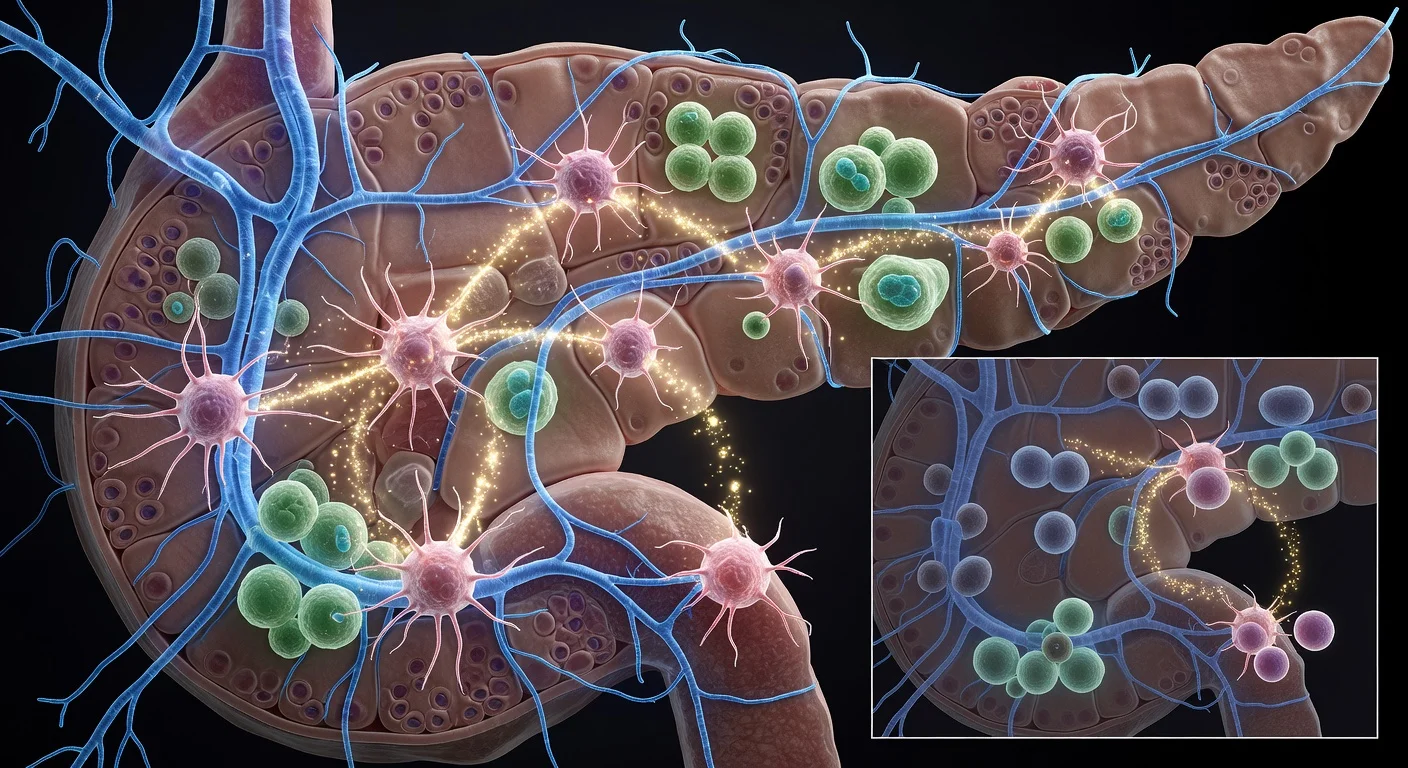
Researchers in Brazil have uncovered how pancreatic cancer uses a protein called periostin to invade nerves and spread early. This discovery explains the disease's aggressiveness and suggests new treatment targets. The findings, published in Molecular and Cellular Endocrinology, highlight the tumor's ability to remodel surrounding tissue.
Pancreatic cancer remains one of the deadliest malignancies, with a death rate nearly matching its diagnosis rate. Worldwide, it claims about 510,000 lives annually from roughly the same number of new cases. In Brazil, the National Cancer Institute estimates 11,000 new diagnoses and 13,000 deaths each year. The most common type, adenocarcinoma, arises in glandular cells and accounts for 90% of cases.
A study led by Carlos Alberto de Carvalho Fraga at the Center for Research on Inflammatory Diseases in São Paulo reveals that tumors do not spread in isolation. Instead, they reprogram nearby healthy tissue using periostin, produced by stellate cells in the pancreas. This protein reshapes the extracellular matrix, enabling cancer cells to invade nerves—a process known as perineural invasion. "Perineural invasion is a marker of cancer aggressiveness," explains oncologist Pedro Luiz Serrano Uson Junior, one of the authors.
Perineural invasion allows cancer cells to travel along nerve pathways, facilitating metastasis and causing severe pain. More than half of cases exhibit this early, often undetected until post-surgery biopsy. The tumor environment also triggers a desmoplastic reaction, forming dense fibrous barriers that hinder chemotherapy and immunotherapy. "That's why pancreatic cancer is still so difficult to treat," Uson notes.
Principal investigator Helder Nakaya, a professor at the University of São Paulo, used advanced gene analysis on 24 samples to map this process. "We were able to integrate data from dozens of samples with extremely powerful resolution," Nakaya says. Only about 10% of patients survive five years post-diagnosis.
The research points to periostin as a promising target. Blocking it or stellate cells could prevent early invasion, aligning with precision medicine trends. Clinical trials for periostin antibodies in other cancers may inform pancreatic applications. "This work points to paths that may guide future approaches to treating pancreatic cancer," Nakaya concludes. Uson adds that such therapies could extend to breast and intestinal cancers, where perineural invasion also occurs.