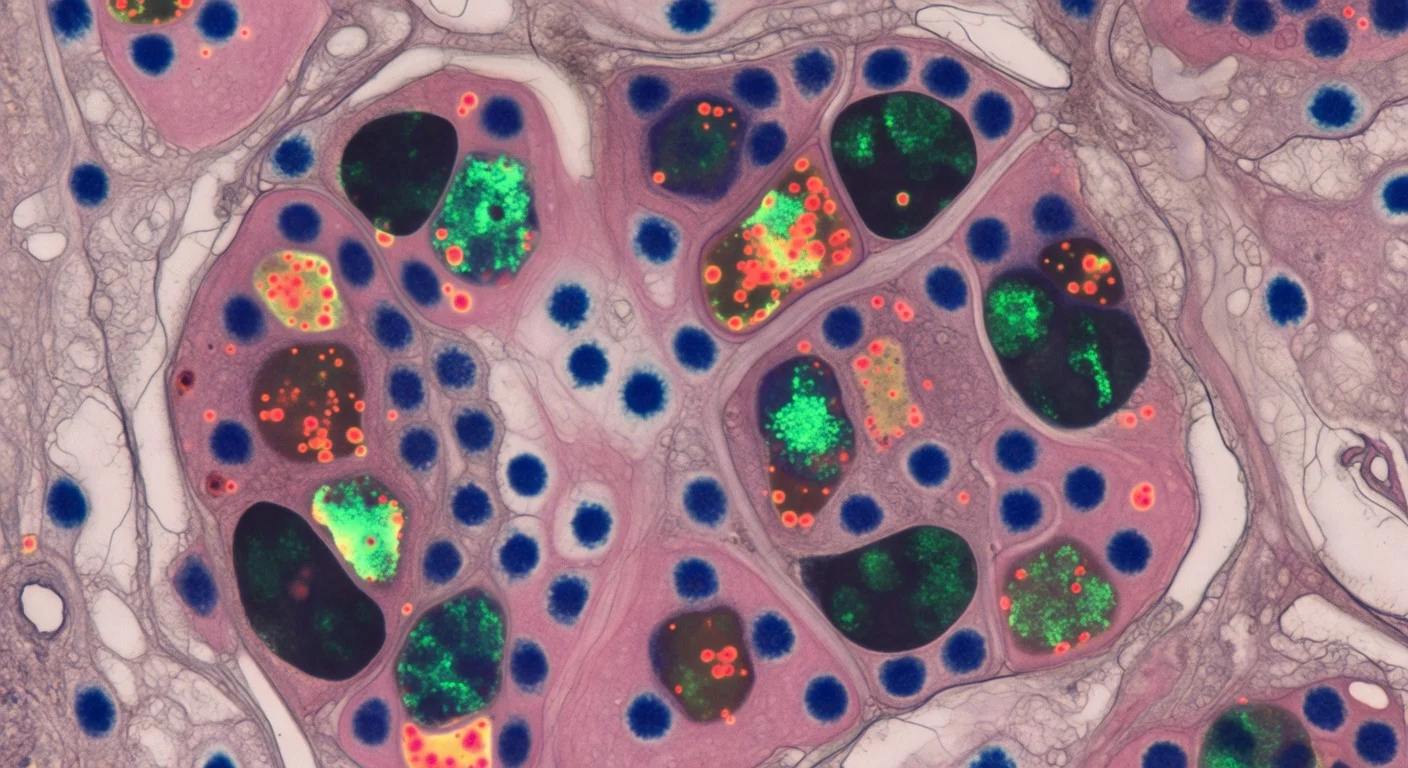
Researchers say they have identified a cellular mechanism that may help explain why PARP inhibitors can affect tumor cells unevenly: in lab-grown slices of human ovarian tumors, some of these drugs accumulated inside lysosomes, forming slow-release stores that created patchy drug distribution across tissue and even between neighboring cells. The findings were reported in a 2026 paper in Nature Communications.
A team led by Dr. Louise Fets at the UK Medical Research Council’s Laboratory of Medical Sciences (LMS) has reported evidence that lysosomes—cell compartments often described as cellular “recycling centers”—can store certain PARP inhibitors inside tumor cells and contribute to uneven drug exposure.
In the study, researchers examined thin slices of ovarian tumor tissue taken from patients and maintained alive outside the body. The explants were treated with PARP inhibitors so the team could track how the drugs moved through intact human tumor tissue rather than standard cell culture models.
To map where the drugs went, the researchers used mass spectrometry imaging to visualize drug molecules within the tissue and paired those measurements with spatial transcriptomics to compare gene activity in areas with higher and lower drug levels from the same tissue section. The maps showed striking variability in drug distribution within individual tumors and between patients, even when the same dose was used.
Dr. Zoe Hall, an associate professor at Imperial College London’s Department of Metabolism, Digestion and Reproduction and a senior author on the work, said the approach enabled direct measurement of drug uptake alongside local gene-expression patterns within the same tumor slice.
The researchers reported that lysosomes played a central role in the patchy distribution. According to the study, some PARP inhibitors were drawn into lysosomes and retained there, creating intracellular “pockets” of drug that could be released over time—effectively acting as slow-release reservoirs that increased exposure in some cells while leaving others with much lower levels.
Not all PARP inhibitors behaved the same way in these experiments. The study found that rucaparib and niraparib were affected by lysosomal storage, while olaparib was not.
Dr. Carmen Ramirez Moncayo, the study’s first author and a postdoctoral researcher at LMS, said the team was surprised by the extent of variability in drug accumulation at the single-cell level and linked that pattern to lysosomal build-up.
Fets said that understanding how drugs are taken up and distributed inside cells could eventually support more tailored therapeutic approaches, including strategies based on the molecular features of a patient’s tumor.
The researchers noted that their work was conducted in tumor tissue maintained outside the body. In patients, drugs are delivered through the bloodstream, and disorganized tumor blood vessels may further influence how evenly drugs spread through tumors. Future work will aim to test these dynamics in animal models and larger patient groups, including relapsed cancers.
The work was supported by the Medical Research Council, Cancer Research UK, and additional funding sources listed by the research team, including a PhD studentship from the Integrative Toxicology Training Partnership administered by the MRC Toxicology Unit and a Victoria’s Secret Global Fund for Women’s Cancers Career Development Award in partnership with Pelotonia and the American Association for Cancer Research (AACR).