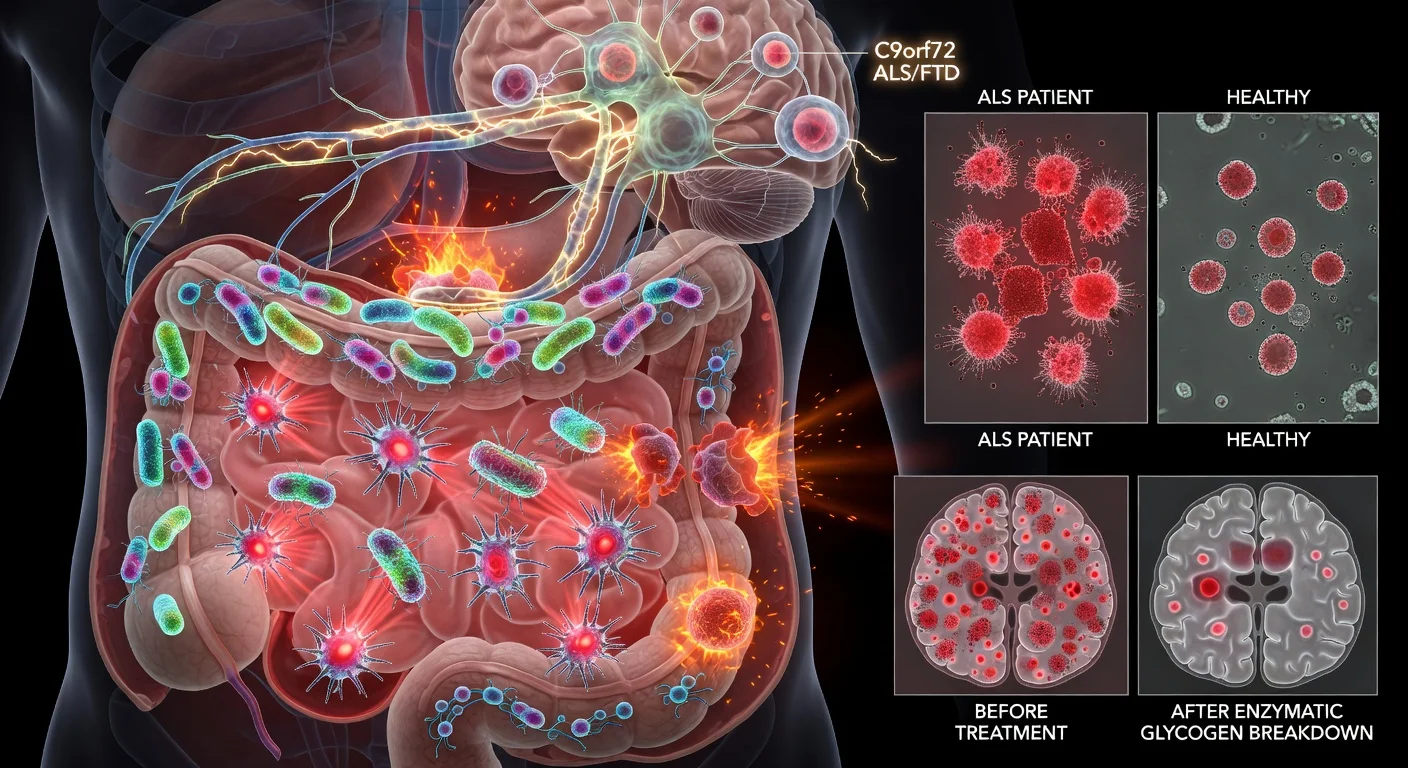
Researchers at Case Western Reserve University report that some gut bacteria can make unusually inflammatory forms of glycogen and that this microbial glycogen can trigger immune activity linked to brain inflammation in models of disease tied to the C9orf72 mutation. In patient stool samples, the team found these glycogen forms more often in ALS and C9orf72-related frontotemporal dementia than in healthy controls, and enzymatically breaking down glycogen in the gut improved outcomes in mice.
Researchers at Case Western Reserve University have reported evidence that certain gut bacteria can produce inflammatory forms of glycogen—a carbohydrate sometimes described as a “sugar”—that can provoke immune responses relevant to amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD), particularly in settings involving the C9orf72 gene.
The study, published in Cell Reports, focused on how microbial products in the gut interact with the immune system in the context of C9orf72, a gene in which a repeat expansion is the most common known genetic cause of ALS and FTD. In a survey of human fecal samples described in the paper, inflammatory forms of glycogen were detected in 15 of 22 ALS samples and in 1 of 1 sample from a patient with C9orf72-associated FTD, compared with 4 of 12 healthy controls.
Aaron Burberry, an assistant professor in the Department of Pathology at the Case Western Reserve School of Medicine and a corresponding author on the work, said the team found that “harmful gut bacteria produce inflammatory forms of glycogen (a type of sugar), and that these bacterial sugars trigger immune responses that damage the brain.”
In experiments using germ-free mice, the researchers reported that colonizing C9orf72-deficient animals with a glycogen-producing bacterial strain (including Parabacteroides merdae in their model) heightened systemic immune changes and signs of central nervous system involvement, including blood–brain barrier disruption and immune-cell infiltration. They also reported that enzymatically digesting glycogen in the gut dampened microglial reactivity in the brain and improved survival in C9orf72-deficient mice.
Alex Rodriguez-Palacios, an assistant professor in the Digestive Health Research Institute at the School of Medicine and a co-author, said the team was able to reduce the harmful glycogen in experiments in ways that “improved brain health and extended lifespan,” according to the university’s summary of the findings.
The researchers argue the work offers a potential explanation for why only some genetically at-risk people develop ALS or FTD: microbial activity in the gut may serve as an environmental factor that influences inflammatory pathways linked to disease. They said the results point to possible therapeutic strategies, including approaches aimed at breaking down inflammatory glycogen in the digestive tract or targeting mechanisms that connect gut microbes to immune activity affecting the nervous system.
Burberry said the group plans larger studies of patient microbiomes and said clinical trials testing whether glycogen degradation could slow ALS/FTD progression “could begin in a year,” according to the university’s release.