Researchers in Brazil report that prolonged use of the proton pump inhibitor (PPI) omeprazole altered iron and calcium measures in adult rats and shifted how several minerals were distributed across organs—changes they say are consistent with higher risks of anemia and possible bone health harms. The authors and Brazil’s health regulator stress that PPIs remain effective for acid-related disorders, but warn against extended, unsupervised use.
A research team in Brazil from the Federal University of São Paulo (UNIFESP) and the ABC Medical School (FMABC) studied how long-term exposure to a common proton pump inhibitor (PPI) affected mineral nutrient bioavailability in rats.
PPIs—such as omeprazole (sold as Prilosec), pantoprazole (Protonix) and esomeprazole (Nexium)—reduce stomach acid by inhibiting the gastric H+,K+-ATPase “proton pump,” and are widely used for conditions including ulcers, gastritis and reflux.
What the researchers did
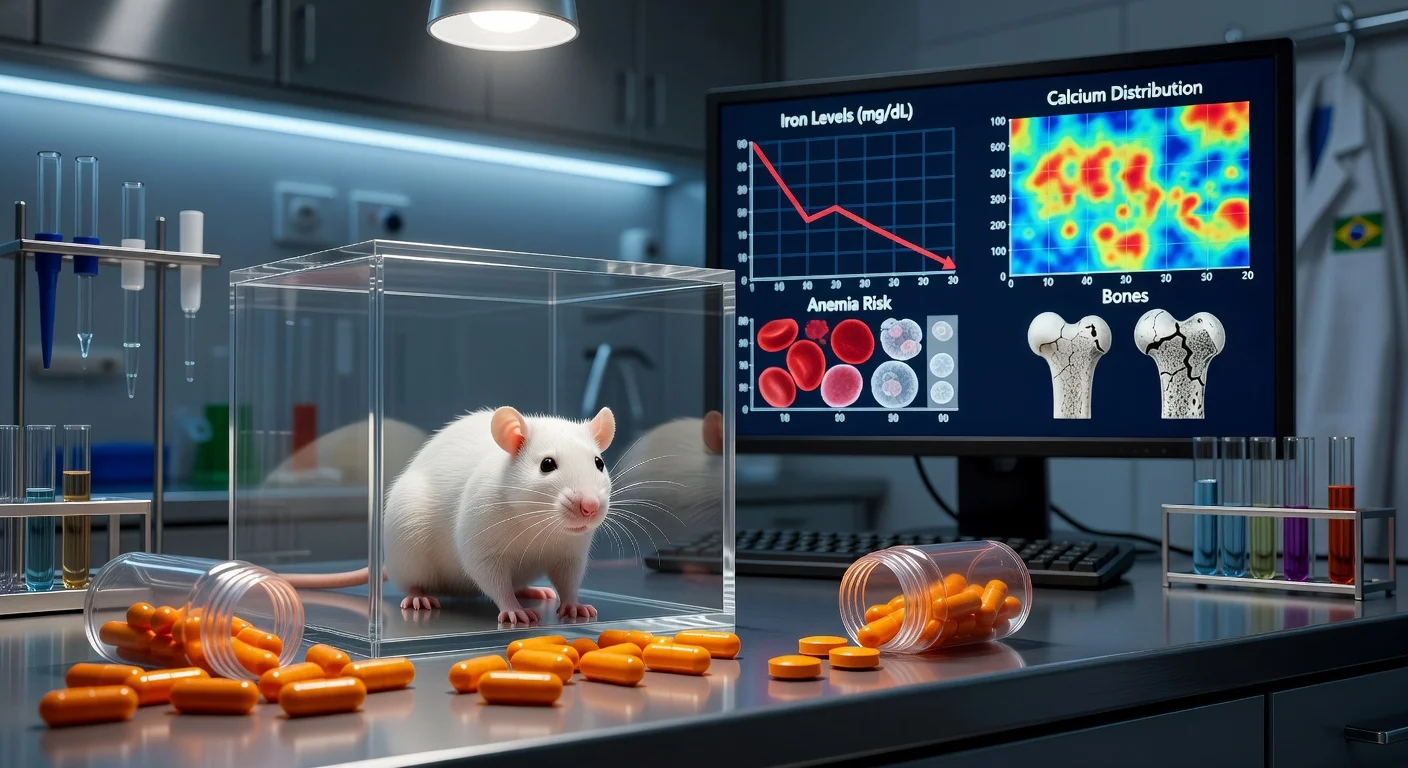
The study, published in ACS Omega in 2025, used adult rats assigned to a control group or to an omeprazole-treated group. Treated animals received omeprazole for 10, 30, or 60 days, after which researchers collected blood and organs for biochemical, hematological and elemental analyses, including mineral quantification using inductively coupled plasma mass spectrometry (ICP‑MS).
What they found
According to the researchers’ report and a research-organization summary, omeprazole exposure was associated with changes in blood and organ mineral measures. They reported higher calcium levels and lower iron levels in blood, along with shifts in mineral distribution across organs—including findings described as mineral buildup in the stomach and altered levels in organs such as the liver and spleen. The paper’s abstract also reports changes in hematological markers, including reductions in red blood cells, hemoglobin and hematocrit, as well as altered leukocyte counts.
The researchers said these patterns are consistent with risks such as microcytic anemia and potential bone-related harms, while emphasizing that the work was conducted in animals and does not establish the same effects in people.
Researchers’ caution on bone risk
“The most worrying finding was the significant increase in calcium in the animals’ bloodstream, which may indicate an imbalance with the removal of the mineral from the bones and a future risk of osteoporosis. However, longer studies are needed to confirm this hypothesis,” said Angerson Nogueira do Nascimento, a UNIFESP professor who coordinated the study with Fernando Fonseca from FMABC.
Whether effects could extend beyond omeprazole
Although the experiment centered on omeprazole, the UNIFESP researchers said other PPIs work through the same mechanism. UNIFESP researcher Andréa Santana de Brito warned that some newer PPIs could potentially have stronger effects because they can act more powerfully and for longer periods. “It isn’t a question of demonizing the drug, which is effective for various gastric conditions. The problem is its trivialized use, even for mild symptoms such as heartburn, and for prolonged periods of months and even years,” she said.
Policy context in Brazil
The researchers pointed to a Brazilian regulatory change that could increase unsupervised use: ANVISA authorized over-the-counter sales of 20 mg omeprazole in November 2025. ANVISA has said the shift is intended to promote “safe and responsible use,” and that limiting non-prescription treatment to a maximum of 14 days is meant to encourage medical evaluation if symptoms persist or recur. The agency also said packages containing more than a 14-day supply cannot be sold without a prescription.
The research team said their findings reinforce the importance of rational PPI use and, when appropriate, individualized medical supervision to assess whether supplementation or other follow-up is needed.